BPC-157 10mg
$68.99
- 1 About BPC-157
- 2 BPC-157 Mechanism of Action
- 3 BPC-157 Research Applications
- 4 BPC-157 vs TB-500 vs GHK-Cu Comparison
- 5 BPC-157 Storage & Handling Guide (Research Use Only)
- 6 Frequently Asked Questions (FAQ)
- 6.1 What purity standard does Research Peptides maintain for BPC-157?
- 6.2 How does Research Peptides verify the identity and purity of BPC-157?
- 6.3 Are Certificates of Analysis (COAs) available for BPC-157 batches?
- 6.4 How should BPC-157 be reconstituted for laboratory research?
- 6.5 What storage conditions help maintain BPC-157 stability?
- 6.6 Is BPC-157 stable in solution once reconstituted?
- 6.7 Why do researchers study BPC-157 alongside peptides such as TB-500 or GHK-Cu?
- 7 Certificate of Analysis (COA) & Quality Assurance
- 8 Scientific References
- 9 Regulatory Status & Legal Considerations (U.S.)
About BPC-157
BPC-157 is a synthetic pentadecapeptide derived from a naturally occurring protective protein fragment originally identified in human gastric juice. Classified as a biologically active peptide fragment, it has drawn scientific interest due to its stability and the broad range of molecular pathways it appears to interact with in experimental systems. In research settings, BPC-157 is commonly investigated as a model compound for studying cellular protection mechanisms, angiogenic signaling, and regulatory pathways associated with tissue integrity and repair processes.
Structurally, BPC-157 is composed of 15 amino acids and belongs to a class of stable peptide fragments that retain biological activity independent of the full-length parent protein. Unlike many naturally occurring peptides that degrade rapidly under physiological conditions, BPC-157 exhibits notable stability in experimental environments [1]. This characteristic makes it a useful tool for researchers examining peptide–cell interactions, signaling cascades, and biochemical pathways where peptide degradation can otherwise complicate experimental reproducibility.
BPC-157 is also examined in controlled laboratory models exploring its interactions with pathways involved in nitric oxide signaling, angiogenesis-related molecular factors, and cytoprotective cellular responses. In vitro and preclinical experimental systems suggest that BPC-157 influences mechanisms related to endothelial function, cellular migration, inflammatory signaling pathways, and gene expression patterns linked to tissue-level stress responses [2]. These experimental contexts make BPC-157 relevant to a variety of biochemical and molecular biology studies focused on cellular resilience and regulatory signaling networks.
The synthetic form of BPC-157 offers meaningful practical advantages for controlled research environments, particularly where experimental consistency and material reliability are critical. Because its molecular composition is defined entirely by the synthesis process rather than biological extraction, researchers can work with a compound of known sequence and predictable behavior. Research Peptide’s rigorous SPPS synthesis and HPLC verification ensures consistent peptide structure across batches, supporting repeatable experiments with reliable, comparable outcomes. Its relatively stable peptide backbone further extends this utility, enabling longer observation windows in experimental models compared with more rapidly degrading peptide fragments.
Every batch undergoes in-house analytical verification by HPLC and Mass Spectrometry, with batch-specific Certificates of Analysis available on product pages or upon request. This commitment to documented traceability directly supports the rigorous sourcing requirements of our institutional customers, including research laboratories and universities.
BPC-157 Mechanism of Action
BPC-157 has been investigated in experimental systems for its ability to influence multiple cellular signaling pathways associated with cytoprotection, angiogenesis, and cellular repair responses. Rather than acting through a single well-defined receptor target, current research suggests the peptide modulates several molecular networks involved in nitric oxide signaling, vascular growth factors, and intracellular survival pathways. These interactions have primarily been observed in cell culture assays and animal-based preclinical models.
Primary Target Binding and Selectivity
The precise molecular binding target of BPC-157 has not been definitively characterized, and research generally indicates that the peptide does not behave as a classical receptor-specific agonist or antagonist. Instead, experimental findings suggest it interacts with regulatory components of endothelial signaling systems and cytoprotective molecular pathways [3].
In endothelial and fibroblast cell models, BPC-157 exposure has been associated with changes in nitric oxide synthase activity and modulation of endothelial signaling regulators. Some experimental studies suggest the peptide may influence endothelial nitric oxide synthase (eNOS) activity and interact with signaling components involved in vascular tone regulation [4].
Because nitric oxide pathways intersect with numerous intracellular signaling cascades, this interaction is often studied as a potential upstream regulatory mechanism for downstream cellular responses.
Downstream Signaling Pathways
Following cellular exposure observed in experimental systems, BPC-157 has been linked to several downstream signaling pathways that regulate cell survival and stress responses. Preclinical studies have reported activation or modulation of pathways such as PI3K/AKT and MAPK/ERK, both of which are commonly involved in cellular growth regulation, survival signaling, and migration processes in cultured cells [5].
Activation of PI3K/AKT signaling in experimental models has been associated with increased phosphorylation of AKT and related survival signaling intermediates. In cell culture assays, this signaling cascade can influence transcriptional programs governing cellular resistance to oxidative stress and apoptotic signaling. Similarly, MAPK/ERK pathway modulation has been observed in endothelial and connective tissue cell systems, where ERK activation is commonly associated with cellular proliferation and migration processes.
Experimental models have also examined BPC-157’s relationship with nitric oxide signaling networks. Nitric oxide acts as a central regulator of vascular and inflammatory signaling, and research suggests the peptide may influence the balance between nitric oxide synthase activity and related signaling mediators. In endothelial assays, this interaction has been linked to altered expression of angiogenesis-related regulatory factors [4].
Angiogenic and Cytoprotective Signaling Networks
Another major focus of BPC-157 research involves molecular pathways associated with angiogenesis and tissue-level stress responses. Experimental studies have reported changes in signaling markers such as vascular endothelial growth factor (VEGF) and related angiogenic regulators in cell-based and animal models. These pathways are frequently investigated in research examining vascular remodeling, endothelial migration, and extracellular matrix interactions.
Cytoprotective signaling pathways are also a central research focus. Laboratory models have examined the peptide’s interaction with inflammatory signaling mediators, oxidative stress pathways, and gene expression networks involved in cellular stress adaptation. For example, experimental systems have explored how BPC-157 exposure may influence transcriptional regulators associated with inflammatory cytokine signaling and cellular recovery processes [5].
Functional Effects Observed in Preclinical Models
In vitro and animal-based research models have documented a range of measurable cellular responses following exposure to BPC-157. These observations include altered endothelial cell migration patterns, modulation of angiogenic biomarker expression, and changes in gene expression associated with inflammatory and oxidative stress pathways.
Experimental studies have also examined the peptide’s effects in models involving vascular injury signaling, connective tissue cellular responses, and nitric oxide-mediated regulatory systems. Researchers frequently evaluate these outcomes using molecular endpoints such as signaling protein phosphorylation levels, cytokine expression profiles, and markers of extracellular matrix remodeling [3][4].
Because many of these pathways intersect with broader cellular regulatory systems, BPC-157 is often studied as a model compound for investigating how short bioactive peptides influence integrated signaling networks involved in cellular resilience and vascular regulation.
Pharmacology-Relevant Design Features
Several structural characteristics make BPC-157 useful for controlled laboratory investigation. The peptide consists of a defined 15-amino-acid sequence that can be reliably reproduced through chemical synthesis, enabling consistent structural identity across research batches. This reproducibility supports repeatable experimental conditions when investigating peptide-mediated signaling mechanisms.
In addition, BPC-157 demonstrates notable stability in experimental environments compared with many endogenous peptide fragments that degrade rapidly in aqueous systems. Studies have reported that the peptide remains relatively resistant to enzymatic breakdown in simulated biological conditions, allowing researchers to observe sustained signaling interactions during extended experimental assays [6].
These signaling systems are highly sensitive, requiring research compounds with exceptional purity and structural consistency to avoid confounding experimental results. Research Peptides produces BPC-157 using advanced automated and manual peptide synthesis platforms that enable precise sequence assembly and carefully controlled purification, consistently achieving purity levels exceeding 99.9%. From synthesis through final packaging, production follows tightly managed laboratory protocols designed to maintain peptide integrity and stability for researchers operating in controlled experimental environments.
BPC-157 Research Applications
BPC-157 is widely examined in laboratory research as a stable peptide fragment used to explore cellular signaling systems associated with tissue integrity, vascular biology, and molecular stress responses. Because the peptide appears to interact with several regulatory pathways simultaneously, it has become a frequently studied compound in experimental models investigating angiogenesis, nitric oxide signaling, inflammatory mediators, and connective tissue cellular dynamics.
In vitro systems and preclinical models commonly use BPC-157 to examine how small bioactive peptides influence cellular signaling networks that regulate endothelial function, extracellular matrix remodeling, and cellular survival pathways. These research applications span biochemical assays, cell culture studies, and animal-based models designed to evaluate molecular signaling changes and measurable physiological markers under controlled laboratory conditions.
Researchers studying peptide signaling, cellular protection mechanisms, and vascular regulatory systems frequently use BPC-157 in experiments to observe how peptide exposure influences transcriptional activity, signaling protein phosphorylation, cytokine expression patterns, and cellular migration processes.
Angiogenesis and Vascular Signaling Research
One of the most common experimental contexts for BPC-157 involves the study of angiogenesis-related signaling pathways. Angiogenesis, the formation of new blood vessels from existing vascular structures, is regulated by complex interactions between endothelial cells, growth factors, and signaling molecules such as vascular endothelial growth factor (VEGF) [7].
Laboratory studies have reported that BPC-157 exposure in endothelial cell cultures can influence signaling pathways associated with vascular growth and endothelial cell migration. Researchers often evaluate molecular endpoints such as VEGF expression levels, endothelial nitric oxide synthase activity, and phosphorylation patterns in signaling proteins associated with vascular development pathways.
Cell culture experiments frequently use endothelial cell lines to observe how peptide exposure affects migration and tube formation assays, which are standard experimental methods used to model angiogenesis in vitro. These studies may also measure changes in transcriptional markers related to vascular growth signaling and extracellular matrix interactions.
Animal-based preclinical research has further examined how BPC-157 interacts with vascular signaling systems in models designed to evaluate blood vessel adaptation, endothelial repair signaling, and regulatory pathways involved in maintaining vascular integrity [4].
Such experimental approaches allow researchers to study how short peptide fragments interact with vascular regulatory networks that influence endothelial signaling and angiogenic responses.
Nitric Oxide Pathway and Endothelial Function Models
Nitric oxide signaling plays a central role in regulating vascular tone, endothelial communication, and cellular stress responses. For this reason, nitric oxide pathways are a major focus in many BPC-157 laboratory studies.
Experimental research has suggested that BPC-157 may influence the nitric oxide system through interactions with nitric oxide synthase activity and related signaling regulators. In vitro experiments using endothelial and fibroblast cell models often measure nitric oxide production levels, enzyme activity changes, and associated signaling protein responses after peptide exposure [4].
Researchers frequently evaluate biochemical endpoints such as nitric oxide metabolites, endothelial nitric oxide synthase expression, and phosphorylation states of regulatory signaling proteins associated with nitric oxide production pathways.
Animal models have also been used to explore how BPC-157 influences nitric oxide–related signaling cascades in physiological systems involving vascular function and inflammatory regulation. These studies often assess molecular markers connected to endothelial signaling stability and nitric oxide–mediated cellular responses.
Because nitric oxide pathways intersect with multiple cellular signaling systems, these research models provide insights into how peptides may influence broader regulatory networks governing vascular biology and cellular stress adaptation.
Connective Tissue and Extracellular Matrix Signaling Studies
Another important research area involving BPC-157 focuses on cellular pathways associated with connective tissue signaling and extracellular matrix regulation. The extracellular matrix is composed of structural proteins and signaling molecules that provide support and regulatory cues for cells within tissues [5].
In laboratory models, researchers frequently examine how peptide exposure influences fibroblast activity, extracellular matrix protein expression, and cellular migration processes involved in tissue structural maintenance. Fibroblast cultures are commonly used to evaluate gene expression patterns related to collagen synthesis, matrix remodeling enzymes, and regulatory growth factors.
Experimental systems may measure molecular indicators such as collagen-associated gene expression, enzyme activity related to matrix remodeling, and signaling pathways that regulate cellular proliferation and structural protein synthesis.
Preclinical animal studies have further explored how BPC-157 interacts with connective tissue signaling systems by measuring histological markers, biochemical indicators of matrix metabolism, and transcriptional changes associated with cellular stress adaptation.
These studies help researchers understand how peptide-mediated signaling may influence cellular communication within structural tissue environments.
Inflammatory Signaling and Cellular Stress Response Research
Inflammatory signaling networks and oxidative stress pathways are also commonly investigated using BPC-157 in experimental systems. Cellular stress responses involve complex interactions between cytokine signaling molecules, oxidative stress regulators, and intracellular survival pathways.
In vitro experiments frequently measure how peptide exposure influences cytokine expression patterns, inflammatory signaling proteins, and markers associated with oxidative stress responses. Macrophage cultures, fibroblast models, and epithelial cell systems are commonly used to evaluate these pathways in controlled research environments [8].
In this context, researchers may examine molecular endpoints such as tumor necrosis factor signaling markers, interleukin expression profiles, and transcriptional regulators involved in inflammatory pathway activation.
Preclinical models have further evaluated biochemical indicators related to cellular stress responses, including enzyme activity changes and signaling proteins involved in apoptosis regulation and cellular survival mechanisms.
These experimental frameworks allow researchers to explore how small bioactive peptides interact with molecular systems responsible for regulating inflammatory signaling and cellular resilience.
Experimental Models of Cellular Migration and Tissue Signaling
Cellular migration and tissue-level signaling are additional research contexts in which BPC-157 is frequently studied. Cellular migration plays a critical role in processes such as vascular remodeling, connective tissue organization, and cellular communication during stress responses [5].
In laboratory experiments, researchers often use scratch assays and migration assays in cultured cell systems to observe how peptides influence cellular movement and signaling coordination between neighboring cells. These assays help evaluate changes in signaling pathways related to cytoskeletal organization, adhesion proteins, and growth factor regulation.
Molecular analyses may include gene expression profiling, phosphorylation markers for signaling proteins involved in migration pathways, and transcriptional responses related to extracellular matrix interactions.
Animal models have also examined how peptide exposure influences cellular signaling patterns involved in tissue organization and vascular regulatory systems. Researchers measure outcomes such as histological tissue markers, molecular signaling indicators, and changes in biochemical pathways associated with cellular communication networks.
These studies help investigators explore how peptide signaling integrates with the broader molecular systems governing cellular coordination and structural tissue dynamics.
| Note: BPC-157 is supplied strictly for laboratory research use only. It’s not approved nor intended for human or veterinary use. |
BPC-157 vs TB-500 vs GHK-Cu Comparison
Researchers frequently evaluate BPC-157 alongside other peptides involved in tissue signaling, vascular biology, and extracellular matrix regulation. Comparing structurally or functionally related compounds can help investigators select appropriate experimental tools depending on the biological pathway of interest, the signaling mechanisms being studied, and the model systems used.
The table below highlights key distinctions between BPC-157 and two commonly referenced peptides in adjacent research domains.
| Field | BPC-157 | TB-500 (Thymosin Beta-4 Fragment) | GHK-Cu |
| Compound Type | Synthetic pentadecapeptide derived from a gastric protein fragment | Synthetic peptide fragment derived from Thymosin Beta-4 | Endogenous copper-binding tripeptide complex |
| Amino Acid Length / Structure | 15–amino acid peptide fragment | 43–amino acid peptide (Thymosin Beta-4 derivative fragment) | Tripeptide (Gly-His-Lys) complexed with copper |
| Primary Biological Systems Studied | Nitric oxide signaling, vascular regulation, cytoprotective pathways | Actin cytoskeleton regulation, cellular migration pathways | Copper-dependent enzymatic signaling and extracellular matrix regulation |
| Key Molecular Pathways Investigated | Nitric oxide synthase signaling, VEGF-related angiogenic pathways, PI3K/AKT and MAPK signaling in experimental models | Actin polymerization dynamics, cytoskeletal remodeling, cellular motility signaling | Copper transport pathways, oxidative stress regulation, extracellular matrix gene expression |
| Typical Research Models | Endothelial cell assays, fibroblast cultures, connective tissue models, rodent preclinical systems | Cell migration assays, angiogenesis assays, connective tissue models, rodent preclinical systems | Fibroblast cultures, biochemical copper signaling assays, extracellular matrix models |
| Mechanistic Research Focus | Multi-pathway signaling modulation affecting vascular and cytoprotective regulatory systems | Cytoskeletal organization and cellular migration signaling | Trace metal–mediated gene regulation and enzyme activation pathways |
| Experimental Stability Characteristics | Relatively stable synthetic peptide fragment resistant to rapid enzymatic degradation in experimental systems | Larger peptide fragment with actin-binding properties studied in cell migration models | Stability dependent on copper coordination and cellular metal transport dynamics |
| Research Context Overlap | Frequently studied in vascular signaling and tissue signaling research | Often studied in tissue signaling and cellular migration models | Commonly examined in extracellular matrix signaling and oxidative balance research |
Researchers often compare BPC-157 with peptides such as TB-500 and GHK-Cu when examining experimental models related to tissue signaling, cellular migration, and extracellular matrix regulation. In research literature, BPC-157 is typically investigated for its interaction with nitric oxide signaling pathways and vascular regulatory systems, while TB-500 studies more commonly focus on actin dynamics and cell migration mechanisms.
GHK-Cu, by contrast, is frequently used as a model copper-binding peptide for exploring gene expression changes and copper-dependent enzymatic signaling. These differences mean researchers studying BPC-157 research applications, TB-500 peptide signaling, or GHK-Cu copper peptide pathways may select different compounds depending on whether their experimental focus involves vascular signaling, cytoskeletal regulation, or trace metal–dependent molecular systems.
BPC-157 Storage & Handling Guide (Research Use Only)
BPC-157 supplied by Research Peptides should be handled like any other laboratory research compound with limited clinical characterization. As with other experimental peptides, appropriate care should be taken to minimize contamination, degradation, or accidental exposure during laboratory work.
Accordingly, all handling should occur within controlled research environments and follow established institutional safety procedures and documentation protocols.
In particular, peptide stability and experimental reproducibility can be influenced by environmental conditions, so researchers should implement standardized handling and storage practices. The following guidance outlines widely accepted laboratory practices for managing research peptides such as BPC-157.
Standard Laboratory Handling Practices
Handling experimental peptides requires consistent laboratory procedures to reduce the risk of contamination and maintain compound integrity during experimental work. The practices below reflect common safety measures used when working with biologically active research compounds in controlled laboratory settings.
- Follow institutional standard operating procedures (SOPs) and approved research protocols when handling research compounds or preparing experimental solutions.
- Use appropriate personal protective equipment (PPE), including a laboratory coat, disposable nitrile or latex gloves, and protective eyewear. Additional protective equipment may be required depending on the research environment or institutional guidelines.
- Conduct weighing, transfer, or solution preparation inside appropriate engineering controls such as a chemical fume hood or biosafety cabinet. These controls help reduce the risk of aerosolization and unintended exposure.
- Use sterile laboratory tools, calibrated pipettes, and clean preparation surfaces to minimize contamination during sample preparation and experimental workflows.
- Clearly label all vials, aliquots, and working solutions with compound name, concentration, preparation date, and lot number to support accurate laboratory documentation.
Consistent adherence to these handling procedures helps maintain experimental reliability and supports safe laboratory operation when working with research peptides.
Note that, BPC-157 is a short peptide fragment, which may make it more susceptible to degradation. This compound is typically supplied in lyophilized (freeze-dried) form, which enhances long-term stability during storage and transportation. However, exposure to moisture prior to reconstitution can promote gradual peptide degradation, so vials should remain sealed until preparation begins.
Preparing fresh solutions or aliquoting reconstituted material can help maintain experimental consistency. For the same reason, gentle mixing is recommended when dissolving lyophilized peptide material, because excessive agitation or vortexing may introduce bubbles or contribute to peptide instability in solution.
Storage Guidelines for Peptide Stability
Proper storage conditions are critical for preserving peptide integrity and ensuring that research materials remain suitable for experimental use. Environmental factors such as temperature, moisture, and light exposure can influence peptide stability over time.
- Store lyophilized BPC-157 in a sealed vial at −4°F (−20°C) for long-term storage. Maintaining a dry, low-temperature environment helps preserve the peptide’s structural integrity.
- Protect stored vials from moisture and humidity by keeping them sealed in desiccated containers or laboratory-grade storage systems.
- Avoid direct exposure to light during storage, particularly for extended periods, as certain peptides may experience gradual degradation when exposed to light.
- After reconstitution, prepared peptide solutions are commonly stored under refrigeration at 36–46°F (2–8°C) for short-term laboratory use.
- When preparing working solutions, researchers may divide the solution into small aliquots to minimize repeated freeze–thaw cycles.
Maintaining controlled storage conditions helps preserve peptide stability and supports reproducible results in downstream experimental systems.
Documentation and Laboratory Traceability
Accurate documentation and traceability are essential when working with research compounds. Maintaining detailed records ensures that experimental materials can be verified and linked to analytical testing results when necessary.
- Record batch numbers and lot identifiers for all peptide materials used in laboratory experiments.
- Retain associated Certificates of Analysis (COAs) and analytical documentation supplied with each batch.
- Document preparation procedures, solvent systems used for reconstitution, and final working concentrations.
- Maintain clear records of storage conditions, freeze–thaw events, and aliquot preparation to ensure experimental consistency.
- Follow institutional record-keeping requirements and regulatory guidance when storing and documenting experimental materials.
Careful documentation practices support reproducibility in laboratory research and help maintain traceability of compounds used across experimental workflows.
This compound is supplied strictly for laboratory research use only and is not intended for human or veterinary use. Research Peptides does not provide medical, diagnostic, or therapeutic guidance.
Frequently Asked Questions (FAQ)
What purity standard does Research Peptides maintain for BPC-157?
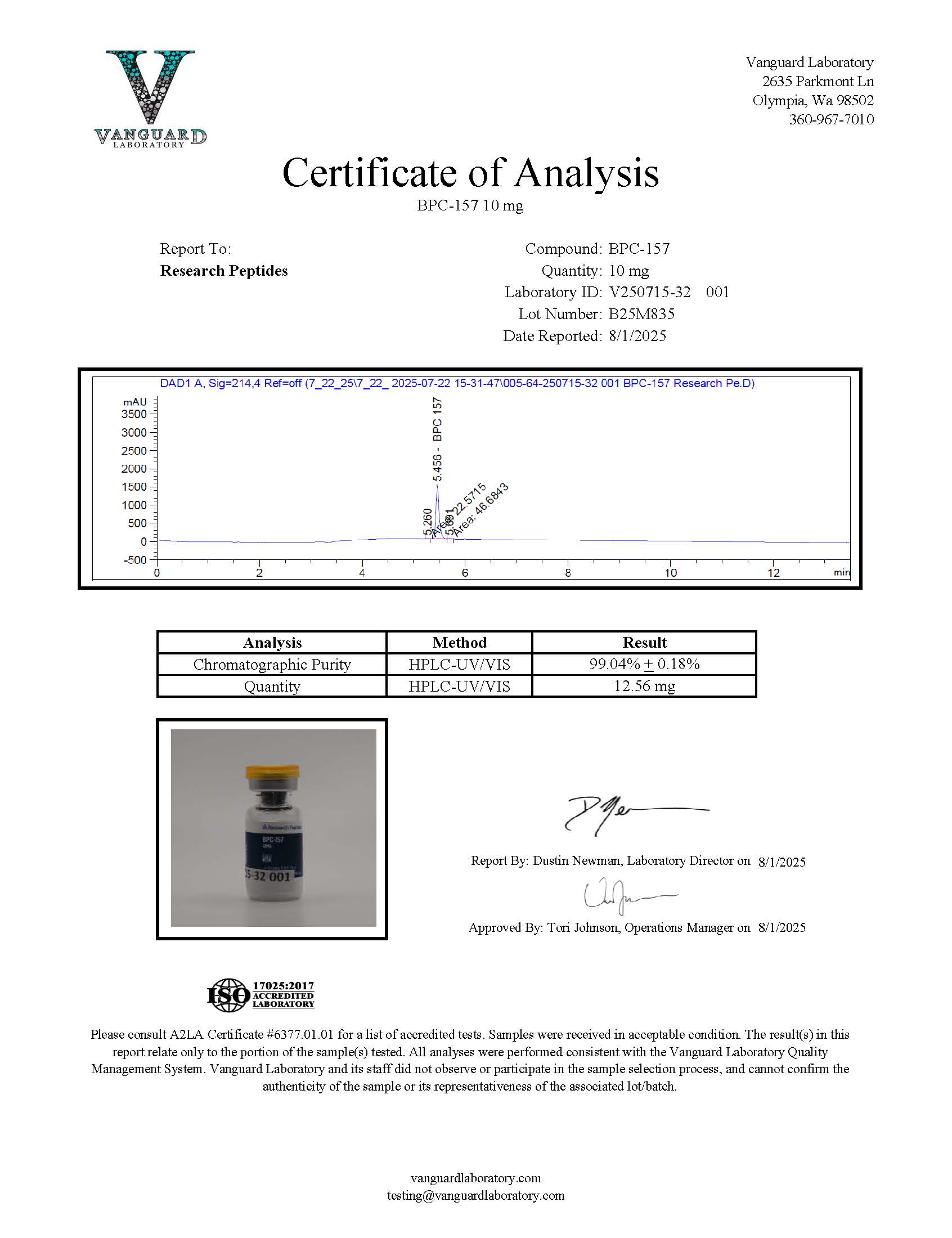
BPC-157 supplied by Research Peptides is produced using advanced automated and manual peptide synthesis systems and purified to research-grade purity levels exceeding 99.9%. Each batch undergoes analytical verification to confirm peptide identity and structural integrity. High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry are used within the company’s in-house analytical laboratory to ensure the compound meets strict quality standards required for reliable experimental research.
How does Research Peptides verify the identity and purity of BPC-157?
Research Peptides conducts multi-stage analytical testing throughout the production process to verify peptide composition and molecular structure. This includes High-Performance Liquid Chromatography (HPLC) to evaluate purity and Mass Spectrometry to confirm molecular identity. These analyses are performed in the company’s in-house analytical laboratory to ensure each batch meets established research-grade specifications before packaging and distribution.
Are Certificates of Analysis (COAs) available for BPC-157 batches?
Yes. Research Peptides provides batch-specific Certificates of Analysis documenting key analytical data used to verify compound identity and purity. These reports typically include results from HPLC purity analysis and Mass Spectrometry verification. Maintaining access to COAs helps researchers confirm the analytical characteristics of each batch and supports traceability for laboratory documentation and experimental reproducibility.
How should BPC-157 be reconstituted for laboratory research?
Researchers commonly reconstitute lyophilized BPC-157 using sterile laboratory-grade solvents such as bacteriostatic water or sterile water for injection, depending on the experimental protocol. The solvent is typically added slowly along the vial wall to reduce foaming and support gradual dissolution of the peptide. Gentle swirling is generally recommended rather than vigorous shaking to help maintain peptide stability during preparation.
What storage conditions help maintain BPC-157 stability?
Lyophilized BPC-157 is typically stored under frozen conditions at approximately −4°F (−20°C) to maintain long-term stability. Vials should remain sealed and protected from moisture and direct light exposure. After reconstitution, prepared solutions are often stored under refrigeration at 36–46°F (2–8°C) for short-term laboratory use. Maintaining controlled storage conditions helps preserve peptide integrity and experimental consistency.
Is BPC-157 stable in solution once reconstituted?
Like many short peptide fragments, BPC-157 may gradually degrade when stored in aqueous solution for extended periods. Researchers often prepare working solutions shortly before use or divide reconstituted solutions into aliquots to minimize repeated freeze–thaw cycles. Refrigerated storage conditions may slow degradation, but stability in solution ultimately depends on solvent composition, storage temperature, and laboratory protocol.
Why do researchers study BPC-157 alongside peptides such as TB-500 or GHK-Cu?
BPC-157 is frequently evaluated alongside peptides such as TB-500 and GHK-Cu because these compounds are studied in related experimental contexts involving tissue signaling, extracellular matrix regulation, and cellular stress responses. While BPC-157 research often focuses on nitric oxide signaling and vascular regulatory pathways, TB-500 studies frequently examine cellular migration and cytoskeletal dynamics, and GHK-Cu research commonly investigates copper-dependent signaling and gene expression pathways. These overlapping but distinct mechanisms make them useful comparative tools in laboratory research models.
Certificate of Analysis (COA) & Quality Assurance
Each lot of BPC-157 supplied by Research Peptides is accompanied by a Certificate of Analysis (COA) intended to support traceability, transparency, and experimental reproducibility in laboratory research. These analytical documents provide verification of key compound characteristics prior to distribution. Access to batch-level documentation allows researchers to confirm the identity and analytical profile of the material when incorporating BPC-157 into controlled experimental workflows.
A Certificate of Analysis serves as a reference record for the analytical testing performed during quality evaluation. The documentation helps researchers verify that each production lot meets established internal quality standards and maintains consistent molecular composition across batches.
A typical COA for BPC-157 peptide materials may include the following analytical information:
- Identity confirmation using Mass Spectrometry or comparable analytical techniques to verify the molecular structure and peptide sequence profile.
- Purity analysis, commonly determined through High-Performance Liquid Chromatography (HPLC), which provides chromatographic data demonstrating the relative purity of the peptide preparation.
- Molecular weight confirmation and related physicochemical characteristics associated with the peptide’s analytical profile.
- Visual and physical description of the lyophilized peptide material, including appearance and form prior to reconstitution.
- Lot identification details, including batch number, testing date, and analytical methods used during the quality verification process.
- Documentation of laboratory testing procedures performed within the Research Peptides analytical workflow, including analytical verification steps used during production and quality assessment.
These analytical records help ensure that researchers can review the characterization data associated with each production lot and maintain proper documentation within laboratory research records.
Certificates of Analysis are available in PDF format for review on the product page or upon request. This documentation allows researchers to verify analytical testing results and confirm the quality verification associated with each batch of BPC-157 supplied by Research Peptides.
Scientific References
- Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review. Vasireddi N, Hahamyan H, Salata MJ, Karns M, Calcei JG, Voos JE, Apostolakos JM. HSS Journal. 2025 Jul 31:15563316251355551. Epub ahead of print.
https://doi.org/10.1177/15563316251355551 - Multifunctionality and Possible Medical Application of the BPC 157 Peptide—Literature and Patent Review. Józwiak M, Bauer M, Kamysz W, Kleczkowska P. Pharmaceuticals. 2025;18(2):185.
https://doi.org/10.3390/ph18020185 - Gastric Pentadecapeptide BPC 157 Resolves Major Vessel Occlusion Disturbances, Ischemia-Reperfusion Injury Following Pringle Maneuver, and Budd-Chiari Syndrome. Sikiric P, Skrtic A, Gojkovic S, Krezic I, Zizek H, Lovric E, Sikiric S, Knezevic M, Strbe S, Milavic M, Kokot A, Blagaic AB, Seiwerth S. World Journal of Gastroenterology. 2022;28(1):23–46.
https://doi.org/10.3748/wjg.v28.i1.23 - Modulatory Effects of BPC 157 on Vasomotor Tone and the Activation of Src-Caveolin-1-Endothelial Nitric Oxide Synthase Pathway. Hsieh MJ, Lee CH, Chueh HY, Chang GJ, Huang HY, Lin Y, Pang JS. Scientific Reports. 2020;10(1):17078.
https://doi.org/10.1038/s41598-020-74022-y - Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing. McGuire FP, Martinez R, Lenz A, Skinner L, Cushman DM. Current Reviews in Musculoskeletal Medicine. 2025;18(12):611–619.
https://doi.org/10.1007/s12178-025-09990-7 - Concerning BPC-157, a Natural Pentadecapeptide, that Acts as a Cytoprotectant and is Believed to Protect the Gastro-Intestinal Tract (GIT). Whitehouse M. Inflammopharmacology. 2025;33(8):4879–4881.
https://doi.org/10.1007/s10787-025-01882-z - BPC 157 Therapy: Targeting Angiogenesis and Nitric Oxide’s Cytotoxic and Damaging Actions, but Maintaining, Promoting, or Recovering Their Essential Protective Functions. Comment on Józwiak et al. Multifunctionality and Possible Medical Application of the BPC 157 Peptide—Literature and Patent Review. Sikiric P, Seiwerth S, Skrtic A, Staresinic M, Strbe S, Vuksic A, Sikiric S, Bekic D, Soldo D, Grizelj B, Novosel L, Beketic Oreskovic L, Oreskovic I, Stupnisek M, Boban Blagaic A, Dobric I. Pharmaceuticals. 2025;18(10):1450.
https://doi.org/10.3390/ph18101450 - Multifunctionality and Possible Medical Application of the BPC 157 Peptide—Literature and Patent Review. Józwiak M, Bauer M, Kamysz W, Kleczkowska P. Pharmaceuticals. 2025;18(2):185.
https://doi.org/10.3390/ph18020185
Regulatory Status & Legal Considerations (U.S.)
BPC-157 is supplied strictly for laboratory research use only. This material is not approved by the U.S. Food and Drug Administration (FDA) for medical, diagnostic, or therapeutic use. It is intended solely for use by qualified professionals in controlled laboratory environments conducting in vitro studies, analytical testing, or preclinical research.
This product is not sold as a drug, dietary supplement, or food product, and it is not intended for human or veterinary use. Researchers and purchasing institutions are responsible for ensuring that all handling, storage, and experimental use of this compound complies with applicable federal, state, and institutional regulations governing laboratory research materials.
Additional information
| CAS: | 137525-51-0 |
|---|---|
| PubChem CID | 9941957 |
| Molecular Weight | 1419.54 g/mol |
| Amino Acid Sequence | Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val |
| PubChem Link | |
| Product Note | For laboratory use only. Not for human or veterinary use. Proper handling and storage (-20°C) are required to maintain stability. Ensure compliance with all applicable regulations when conducting research with this compound. Peptides will arrive in a lyophilized (powder) form for maximum stability. |