CJC-1295 No DAC 5mg
$51.99
- 1 About CJC-1295 No DAC
- 2 CJC-1295 No DAC Mechanism of Action
- 3 CJC-1295 No DAC Research Applications
- 4 CJC-1295 No DAC vs Sermorelin vs Ipamorelin Comparison
- 5
- 6 CJC-1295 No DAC Storage & Handling Guide (Research Use Only)
- 7 Frequently Asked Questions (FAQ)
- 7.1 What purity standard does Research Peptides maintain for CJC-1295 No DAC?
- 7.2 Are Certificates of Analysis available for CJC-1295 No DAC?
- 7.3 How should CJC-1295 No DAC be reconstituted for laboratory research?
- 7.4 What storage conditions help maintain CJC-1295 No DAC stability?
- 7.5 Is CJC-1295 No DAC stable in aqueous solution?
- 7.6 What makes CJC-1295 No DAC useful in peptide signaling research?
- 7.7 Why do researchers compare CJC-1295 No DAC with peptides like Sermorelin or Ipamorelin?
- 7.8 What structural characteristics distinguish CJC-1295 No DAC from native GHRH fragments?
- 8 Certificate of Analysis (COA) & Quality Assurance
- 9 Scientific References
- 10 Regulatory Status & Legal Considerations (U.S.)
About CJC-1295 No DAC
CJC-1295 No DAC, commonly referred to as Modified GRF (1-29), is a synthetic peptide analogue of growth hormone–releasing hormone (GHRH). The compound was developed to mimic the biological signaling activity of endogenous GHRH while offering improved stability for laboratory study. In research environments, CJC-1295 No DAC is frequently used as a molecular tool for investigating neuroendocrine signaling pathways associated with the growth hormone axis [1].
CJC-1295 No DAC is derived from earlier research on growth hormone–releasing hormone (GHRH) analogues conducted in the 1990s and early 2000s by peptide researchers investigating ways to stabilize the active fragment of human GHRH. The compound is based on the biologically active region of GHRH (1–29), with several strategic amino acid substitutions designed to improve resistance to enzymatic degradation.
Modified GRF (1-29), the peptide that later became associated with CJC-1295 variants, emerged from this work as a stable research analogue for studying GHRH receptor signaling and growth hormone axis regulation in experimental models.
Structurally, CJC-1295 No DAC retains the first 29 amino acids of endogenous GHRH, preserving the N-terminal region critical for GHRH receptor binding and activation. Unlike longer-acting conjugated variants, this analogue lacks a drug affinity complex (DAC), resulting in a shorter activity profile that more closely approximates natural pulsatile GH release patterns. This distinction makes it particularly useful in studies requiring precise temporal control of GH axis stimulation [2].
Because of its structural relationship to endogenous GHRH, CJC-1295 No DAC is commonly explored in laboratory studies examining GHRH receptor signaling, intracellular cAMP pathways, and regulatory mechanisms within the hypothalamic–pituitary axis. Researchers may also use the peptide in controlled in-vitro systems, cellular assays, and preclinical models designed to evaluate peptide-receptor binding dynamics, downstream gene expression, and hormone signaling cascades [3].
CJC-1295 No DAC synthesized via rigorously controlled production has a consistent and reproducible peptide structure, making it well suited for research compared to the endogenous GHRH. Its modified sequence helps maintain molecular stability while preserving receptor-binding characteristics that allow detailed analysis of GHRH-related pathways in laboratory settings.
Research Peptides supplies CJC-1295 No DAC (5mg) as a high-purity research compound manufactured using modern peptide synthesis and purification methods. Each batch undergoes analytical verification, with batch-specific Certificates of Analysis available on the product pages to support laboratory documentation and experimental reproducibility.
Research-Grade Purity and Analytical Verification
CJC-1295 No DAC (5mg) offered by Research Peptides is produced using advanced peptide synthesis techniques designed to achieve precise sequence assembly and high batch consistency. Our manufacturing processes typically utilizes solid-phase peptide synthesis (SPPS), with additional liquid-phase processes applied where appropriate during peptide construction.
Following synthesis, the peptide undergoes high-resolution purification through preparative high-performance liquid chromatography (HPLC) to isolate the target compound.
Each production batch is subjected to analytical verification to confirm identity and purity. Analytical HPLC testing is used to determine the purity profile, while mass spectrometry is employed to verify molecular weight and confirm correct sequence assembly. Together, these analytical methods help ensure that the peptide meets a purity standard of ≥99% while validating structural integrity for laboratory research use.
To support research documentation and transparency, Research Peptides provides batch-specific Certificates of Analysis (COA) detailing purity results and analytical verification data. As a U.S. supplier focused on research compounds, Research Peptides maintains consistent manufacturing standards and dependable domestic shipping to support laboratories and independent researchers conducting experimental work.
CJC-1295 No DAC Mechanism of Action
CJC-1295 No DAC functions as a synthetic analogue of growth hormone–releasing hormone (GHRH) that interacts with the GHRH receptor (GHRH-R), a G protein–coupled receptor primarily expressed on somatotroph cells of the anterior pituitary. In experimental systems, the peptide acts as a receptor agonist that stimulates intracellular signaling pathways associated with growth hormone regulation [4].
These interactions have been characterized in cell culture studies, receptor-binding assays, and preclinical models investigating neuroendocrine signaling within the hypothalamic–pituitary axis.
Primary Target Binding and Selectivity
CJC-1295 No DAC binds to the GHRH receptor, functioning as a selective agonist that mimics the signaling activity of endogenous GHRH. The GHRH receptor belongs to the class B family of G protein–coupled receptors and mediates hormone signaling through ligand-induced receptor activation [1]. Binding of CJC-1295 No DAC to this receptor has been observed in experimental systems designed to evaluate peptide-receptor interactions and downstream hormone signaling mechanisms.
Because the compound is derived from the active region of human GHRH (1–29), it retains the receptor recognition elements necessary for selective interaction with GHRH-R. This selectivity allows researchers to isolate and study receptor-mediated signaling processes without introducing unrelated receptor activity that could complicate experimental interpretation.
Downstream Signaling Pathways
Upon receptor binding, activation of the GHRH receptor stimulates intracellular signaling pathways primarily associated with cyclic adenosine monophosphate (cAMP). Engagement of the receptor activates adenylate cyclase through Gs protein coupling, increasing intracellular cAMP levels. Elevated cAMP can then activate protein kinase A (PKA), which regulates multiple downstream processes within pituitary cells [2].
In laboratory models, these signaling cascades influence transcription factors involved in growth hormone gene expression and secretion pathways. Additional signaling interactions involving MAPK and ERK pathways have also been observed in experimental studies examining how GHRH receptor activation affects cellular proliferation and gene regulation in endocrine cell systems.
Functional Effects Observed in Preclinical Models
In preclinical and laboratory research, activation of the GHRH receptor by CJC-1295 No DAC has been associated with measurable changes in hormone signaling markers and intracellular pathway activation. Experimental studies have reported increases in cAMP production, modulation of growth hormone–related transcriptional activity, and alterations in downstream signaling proteins within pituitary cell models [5].
Animal studies evaluating GHRH analogues have also documented endocrine signaling responses consistent with receptor activation, providing researchers with models for studying the regulation of the growth hormone axis and its associated biochemical pathways.
Pharmacology-Relevant Design Features
CJC-1295 No DAC incorporates several amino acid substitutions compared with native GHRH (1–29), which help improve resistance to enzymatic degradation while maintaining receptor-binding capability. These structural modifications support greater stability in laboratory assay systems and extend the functional activity of the peptide relative to unmodified GHRH fragments.
Unlike DAC-modified versions of CJC-1295, the No DAC variant does not contain the albumin-binding Drug Affinity Complex. As a result, it is often used in experimental settings where researchers want to evaluate the intrinsic receptor pharmacology of the modified GRF peptide without extended plasma-binding effects that alter activity duration in biological models.
CJC-1295 No DAC Research Applications
CJC-1295 No DAC (Modified GRF 1-29) is widely studied in peptide research as a synthetic analogue of growth hormone–releasing hormone (GHRH). Because it mimics the receptor-binding region of endogenous GHRH while incorporating stabilizing amino-acid substitutions, the peptide provides researchers with a controlled experimental tool for examining neuroendocrine signaling pathways associated with growth hormone regulation.
In laboratory research, CJC-1295 No DAC is commonly used to investigate GHRH receptor pharmacology, intracellular cAMP signaling, and regulatory mechanisms within the hypothalamic–pituitary axis.
In particular, researchers study CJC-1295 No DAC in controlled receptor-binding assays, endocrine cell culture systems, and molecular signaling experiments designed to characterize hormone-regulated pathways. Because the peptide lacks the Drug Affinity Complex (DAC) modification present in other CJC-1295 variants, it allows investigators to evaluate receptor activation dynamics without the confounding influence of albumin-binding extensions that can significantly alter pharmacokinetics in biological systems.
This lack of DAC moiety makes CJC-1295 No DAC particularly useful in mechanistic studies focused on the intrinsic signaling behavior of GHRH analogues.
Across peptide research literature, CJC-1295 No DAC and related modified GRF peptides have also been used in multiple experimental contexts designed to evaluate growth hormone regulatory networks. These include transcriptional profiling studies, endocrine signaling experiments, and preclinical animal models [6].
These studies help researchers better understand how GHRH receptor activation influences intracellular signaling cascades, gene expression patterns, and endocrine communication between the hypothalamus and pituitary gland.
Neuroendocrine Signaling and Growth Hormone Axis Research
One of the primary areas of CJC-1295 No DAC research involves the study of neuroendocrine signaling pathways that regulate the growth hormone axis. In physiological systems, growth hormone release from the anterior pituitary is controlled through complex interactions between hypothalamic hormones, including GHRH and somatostatin. CJC-1295 No DAC functions as a synthetic agonist of the GHRH receptor, allowing researchers to selectively activate this signaling pathway in experimental models [5].
Researchers frequently use pituitary cell cultures or engineered receptor-expression systems to evaluate how GHRH receptor activation influences intracellular signaling processes. Activation of this receptor typically stimulates adenylate cyclase through G-protein coupling, resulting in increased cyclic AMP (cAMP) production. Elevated cAMP levels can then activate protein kinase A (PKA), a signaling enzyme that regulates transcriptional processes associated with hormone synthesis [3].
Experimental endpoints in these studies may include measurement of intracellular cAMP concentrations, phosphorylation of signaling proteins, or expression levels of growth hormone–related transcription factors. These measurements help researchers map how GHRH receptor signaling propagates through intracellular pathways and contributes to endocrine regulation.
Animal models have also been used in preclinical research to study how GHRH analogues influence systemic hormone signaling networks. In such studies, investigators may monitor endocrine biomarkers, receptor expression patterns, or gene transcription changes within hypothalamic and pituitary tissues to better understand the regulatory architecture of the growth hormone axis.
GHRH Receptor Pharmacology and Ligand Binding Studies
Another important area of CJC-1295 No DAC research involves receptor pharmacology and ligand-binding analysis. Because the peptide closely resembles the active segment of endogenous GHRH, it serves as a useful probe for examining how structural modifications affect receptor activation and signaling behavior [7].
Researchers often employ receptor-binding assays using recombinant cell lines engineered to express the human GHRH receptor. In these systems, they can compare the binding affinity of modified peptides such as CJC-1295 No DAC with that of native GHRH fragments. These experiments help clarify which amino-acid substitutions contribute to improved stability, receptor selectivity, or signaling potency.
Typical experimental endpoints in receptor pharmacology studies include ligand-binding affinity measurements, receptor activation curves, and intracellular second-messenger production such as cAMP accumulation. Researchers may also evaluate how peptide modifications influence receptor internalization, desensitization, or recycling within cellular models.
Such studies contribute to a deeper understanding of how peptide-based ligands interact with class B G protein–coupled receptors (GPCRs), a receptor family that includes GHRH-R as well as receptors involved in metabolic and endocrine signaling pathways.
Intracellular Signaling Pathway Research
CJC-1295 No DAC is also studied as a research tool for investigating downstream signaling pathways triggered by GHRH receptor activation. Because this receptor primarily signals through the cAMP-PKA pathway, the peptide is frequently used in molecular experiments designed to map signaling cascades associated with hormone regulation [2].
In cell-based assays, activation of the GHRH receptor by CJC-1295 No DAC can stimulate adenylate cyclase activity, increasing intracellular cAMP levels. This rise in cAMP subsequently activates protein kinase A, which phosphorylates multiple downstream targets involved in gene transcription and cellular signaling.
Researchers may analyze several molecular endpoints to evaluate these processes. Common measurements include phosphorylation markers associated with the PKA pathway, activation of transcription factors such as CREB (cAMP response element–binding protein), and expression of genes involved in endocrine signaling networks.
Additional signaling pathways, including MAPK and ERK pathways, have also been explored in experimental studies of GHRH receptor activation. These pathways are often investigated using biochemical assays that measure kinase activation, gene expression changes, or protein phosphorylation patterns following receptor stimulation.
Through these experiments, scientists can build detailed models describing how peptide ligands influence intracellular communication networks and how those signals propagate within endocrine cells.
Peptide Stability and Structural Optimization Research
A further research application of CJC-1295 No DAC relates to peptide design and structural optimization. Native GHRH fragments are relatively susceptible to enzymatic degradation in biological systems, which limits their utility in experimental models. Modified GRF peptides, including CJC-1295 No DAC, were developed specifically to address this limitation [6].
Researchers studying peptide pharmacology often investigate how targeted amino-acid substitutions influence peptide stability, receptor binding characteristics, and functional signaling activity. In the case of CJC-1295 No DAC, several substitutions within the GRF (1-29) sequence increase resistance to enzymatic cleavage while maintaining the structural features required for receptor activation.
Experimental work in this area frequently involves comparative stability studies in cell culture media or biological fluids, where investigators measure peptide degradation rates using analytical techniques such as high-performance liquid chromatography (HPLC) or mass spectrometry. Structural modeling and receptor-binding experiments may also be used to evaluate how modifications influence peptide conformation and receptor interaction dynamics.
These types of studies contribute to broader peptide engineering research, helping scientists design synthetic analogues with improved stability and predictable signaling characteristics for experimental applications.
Comparative Studies of GHRH Analogues
CJC-1295 No DAC is often included in comparative studies examining different GHRH analogue variants. Researchers may evaluate how modified GRF peptides behave relative to endogenous GHRH fragments or longer-acting analogues that incorporate albumin-binding modifications such as the Drug Affinity Complex (DAC).
Comparative experiments can involve receptor-binding assays, endocrine signaling measurements, or animal models designed to examine how structural modifications influence peptide pharmacology. By studying peptides with and without DAC modifications, researchers can investigate how albumin-binding extensions alter peptide distribution, signaling duration, and receptor interaction profiles in biological systems.
These comparative analyses help researchers better understand how peptide structure influences pharmacological behavior, providing insight into the design of future peptide-based signaling probes used in endocrine and receptor pharmacology research.
| Note: CJC-1295 No DAC supplied by Research Peptides is intended strictly for laboratory research use only and is not approved for human or veterinary use. |
CJC-1295 No DAC vs Sermorelin vs Ipamorelin Comparison
Researchers studying growth hormone–releasing pathways frequently evaluate several peptide ligands that interact with the growth hormone axis. CJC-1295 No DAC, Sermorelin, and Ipamorelin are commonly discussed together in peptide research literature because they influence related neuroendocrine signaling systems through different molecular mechanisms.
The comparison below highlights structural differences, signaling pathways, and experimental research contexts in which these peptides are studied.
| Field | CJC-1295 No DAC | Sermorelin | Ipamorelin |
| Compound Class | Synthetic GHRH analogue (Modified GRF 1-29) | Synthetic GHRH analogue | Growth hormone secretagogue (GHS) peptide |
| Structural Characteristics | 29-amino-acid peptide derived from GHRH with stabilizing substitutions; no DAC albumin-binding modification | 29-amino-acid peptide identical to the active fragment of human GHRH | Short synthetic pentapeptide (5 amino acids) |
| Primary Biological Systems Studied | Hypothalamic–pituitary signaling and GHRH receptor activation | GHRH receptor signaling and endogenous growth hormone regulation | Ghrelin receptor (GHS-R1a) signaling and growth hormone secretagogue pathways |
| Key Molecular Pathways Investigated | GHRH receptor → cAMP / PKA signaling | GHRH receptor → cAMP / PKA signaling | Ghrelin receptor → PLC / Ca²⁺ and growth hormone secretagogue pathways |
| Typical Experimental Models | Pituitary cell cultures, receptor binding assays, endocrine signaling models, preclinical neuroendocrine studies | Cell-based GHRH receptor assays, endocrine cell models, hormone signaling experiments | Receptor pharmacology assays, ghrelin receptor signaling models, metabolic pathway studies |
| Mechanistic Research Focus | Modified GRF analogue designed to improve peptide stability while preserving GHRH receptor interaction | Direct mimic of endogenous GHRH fragment used to study physiological receptor signaling | Selective ghrelin receptor agonist used to study secretagogue signaling pathways |
| Experimental Stability Characteristics | Modified residues improve resistance to enzymatic degradation compared with native GHRH fragments | Native sequence; relatively susceptible to enzymatic degradation in biological systems | Short peptide with relatively stable receptor interaction in experimental assays |
| Research Context Overlap | Neuroendocrine regulation, receptor pharmacology, peptide signaling research | Endocrine signaling and physiological GHRH pathway studies | Growth hormone secretagogue pathway research and receptor pharmacology |
Researchers investigating CJC-1295 No DAC research often focus on how modified GHRH analogues influence receptor signaling dynamics and intracellular cAMP pathways. Because the peptide includes stabilizing substitutions relative to native GHRH fragments, it is frequently used in CJC-1295 peptide studies examining receptor pharmacology and growth hormone–related signaling pathways in cell culture or endocrine models.
By contrast, Sermorelin research typically centers on the natural GHRH (1-29) sequence and its direct interaction with the GHRH receptor in physiological signaling experiments. This makes Sermorelin useful when researchers want to examine endogenous GHRH pathway behavior without structural modifications.
Ipamorelin research, on the other hand, targets a different receptor system entirely: the ghrelin receptor (GHS-R1a). Rather than acting on the GHRH receptor, Ipamorelin activates secretagogue signaling pathways associated with ghrelin-mediated hormone regulation. As a result, Ipamorelin studies are often used to explore alternative mechanisms influencing growth hormone signaling networks.
Because these peptides interact with distinct but overlapping signaling systems, researchers may include multiple compounds in comparative growth hormone axis signaling pathway experiments. Evaluating receptor-specific ligands such as CJC-1295 No DAC, Sermorelin, and Ipamorelin allows investigators to better understand how different molecular signals regulate endocrine communication within neuroendocrine models.
CJC-1295 No DAC Storage & Handling Guide (Research Use Only)
CJC-1295 No DAC should be handled like any other experimental peptide research compound with limited clinical characterization outside controlled research environments. As with other research compounds, appropriate safety procedures and materials-handling protocols should be followed to protect both personnel and compound integrity.
All preparation and handling should occur within regulated laboratory environments operating under established institutional protocols and safety procedures.
Standard Laboratory Handling Practices
Proper handling procedures help reduce contamination risk, preserve compound integrity, and support safe laboratory operations during experimental work with peptide materials.
- Follow institutional standard operating procedures (SOPs) and approved laboratory research protocols when working with peptide compounds
- Wear appropriate personal protective equipment (PPE), including laboratory coats, gloves, and eye protection
- Handle materials within appropriate engineering controls such as chemical fume hoods or biosafety cabinets when preparing solutions
- Use sterile syringes, pipettes, and calibrated laboratory equipment when preparing peptide solutions or dilutions
- Label all aliquots and prepared solutions clearly with compound name, concentration, preparation date, and batch or lot number
- Maintain clear laboratory documentation for compound preparation, storage conditions, and experimental use
Consistent adherence to established laboratory practices supports both experimental reproducibility and safe research operations. Proper labeling, documentation, and controlled handling also help maintain the traceability of research materials across experiments.
Storage Guidelines for Compound Stability
Environmental conditions such as temperature, moisture exposure, and light can influence the stability of peptide compounds and therefore affect experimental reliability.
- Store lyophilized CJC-1295 No DAC at −4°F (−20°C) for long-term storage
- Store reconstituted peptide solutions under refrigeration at 36–46°F (2–8°C) when short-term use is required
- Protect lyophilized material from moisture using tightly sealed containers or desiccated storage environments
- Limit exposure to ambient laboratory light when vials are open or solutions are prepared
- Minimize repeated freeze–thaw cycles by preparing small aliquots suitable for individual experimental runs
Maintaining controlled storage conditions helps preserve peptide structure and reduces degradation risk over time. Careful storage practices are particularly important for maintaining consistent experimental results when working with peptide-based signaling probes.
Compound-Specific Stability and Reconstitution Considerations
CJC-1295 No DAC is a 29–amino acid peptide analogue derived from modified GRF (1–29). Like many research peptides, it may be susceptible to degradation in aqueous environments and should therefore be handled carefully during preparation and storage.
- Reconstitute the peptide using sterile water or bacteriostatic water commonly used in peptide research preparations
- Add solvent slowly along the vial wall and avoid vigorous agitation to reduce peptide foaming or structural stress
- Prepare small aliquots of reconstituted solution when experiments require repeated use to reduce degradation from freeze–thaw cycles
- Avoid prolonged storage of aqueous solutions when possible, as peptide hydrolysis can occur over time
These preparation considerations reflect common stability characteristics observed in synthetic peptide research compounds. Proper reconstitution and aliquoting practices can help maintain structural integrity and support consistent experimental outcomes in peptide signaling studies.
This compound is supplied strictly for laboratory research use only and is not intended for human or veterinary use. Research Peptides does not provide medical, diagnostic, or therapeutic guidance.
Frequently Asked Questions (FAQ)
What purity standard does Research Peptides maintain for CJC-1295 No DAC?
CJC-1295 No DAC supplied by Research Peptides is manufactured to a purity standard of ≥99%. Each batch undergoes analytical verification using high-performance liquid chromatography (HPLC) to evaluate purity profiles and confirm compound consistency. Mass spectrometry is also used to verify molecular identity and sequence integrity, supporting reliable use in laboratory research applications involving peptide signaling and receptor studies.
Are Certificates of Analysis available for CJC-1295 No DAC?
Yes. Research Peptides provides batch-specific Certificates of Analysis (COA) for CJC-1295 No DAC. These documents include analytical data such as HPLC purity results and mass spectrometry identity confirmation. COAs allow researchers to verify compound specifications and maintain proper documentation for laboratory recordkeeping, experimental reproducibility, and quality assurance within regulated research environments.
How should CJC-1295 No DAC be reconstituted for laboratory research?
CJC-1295 No DAC is typically supplied by Research Peptides as a lyophilized peptide powder to maintain stability during storage and transport. In laboratory settings, researchers commonly reconstitute peptides using sterile water or bacteriostatic water. Solvent is generally added slowly along the vial wall, followed by gentle swirling to dissolve the peptide while minimizing agitation that could affect peptide structure.
What storage conditions help maintain CJC-1295 No DAC stability?
For long-term storage, lyophilized CJC-1295 No DAC is generally maintained at −4°F (−20°C). Once reconstituted, peptide solutions are typically stored under refrigeration at 36–46°F (2–8°C) for short-term experimental use. Researchers should prepare aliquots to minimize repeated freeze–thaw cycles, which can contribute to peptide degradation and variability in experimental outcomes.
Is CJC-1295 No DAC stable in aqueous solution?
Like many synthetic peptides, CJC-1295 No DAC may gradually degrade in aqueous solution due to hydrolysis or enzymatic activity. For this reason, researchers commonly prepare only the amount required for near-term experiments and store reconstituted solutions under refrigerated conditions. Aliquoting prepared solutions can help limit repeated freeze–thaw exposure and maintain experimental consistency.
What makes CJC-1295 No DAC useful in peptide signaling research?
CJC-1295 No DAC is widely studied in peptide research because it acts as a synthetic analogue of growth hormone–releasing hormone (GHRH). By activating the GHRH receptor in experimental models, researchers can investigate intracellular signaling pathways such as cAMP and PKA activation. These properties make the peptide useful in studies exploring neuroendocrine signaling and receptor pharmacology.
Why do researchers compare CJC-1295 No DAC with peptides like Sermorelin or Ipamorelin?
Researchers often evaluate CJC-1295 No DAC alongside peptides such as Sermorelin and Ipamorelin when studying growth hormone signaling pathways. While CJC-1295 No DAC and Sermorelin interact with the GHRH receptor, Ipamorelin activates the ghrelin receptor pathway. Comparing these peptides in experimental systems helps researchers examine how different signaling mechanisms influence endocrine regulatory networks.
What structural characteristics distinguish CJC-1295 No DAC from native GHRH fragments?
CJC-1295 No DAC is derived from the active region of human GHRH (1–29) but incorporates several amino-acid substitutions designed to improve stability against enzymatic degradation. These modifications allow the peptide to maintain receptor-binding capability while remaining more resistant to rapid breakdown in experimental systems. This structural design makes it useful for controlled studies of GHRH receptor signaling dynamics.
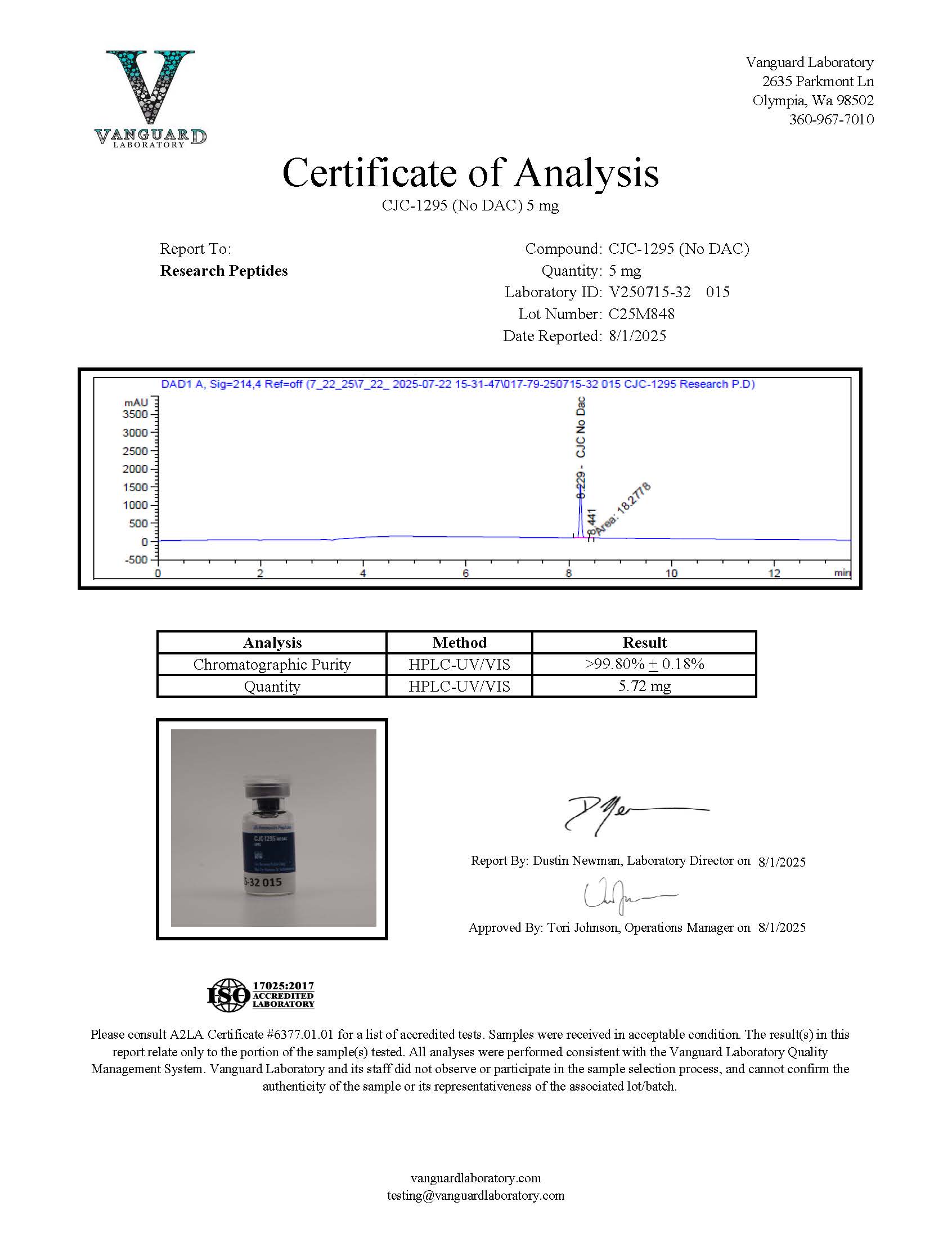
Certificate of Analysis (COA) & Quality Assurance
Each lot of CJC-1295 No DAC sold by Research Peptides is accompanied by a Certificate of Analysis (COA). This document provides analytical documentation associated with the peptide and supports transparency in laboratory research workflows. Analytical verification is performed prior to product release to confirm compound identity and evaluate purity characteristics for each manufactured batch.
For peptide research compounds such as CJC-1295 No DAC, these records help ensure that researchers receive materials that have undergone documented analytical evaluation. The COA provides reference data that laboratories may use when recording compound specifications, verifying experimental inputs, or maintaining internal research documentation.
The Certificate of Analysis serves as a formal record of the analytical testing performed during quality evaluation. These records allow researchers to review verification data related to compound identity, analytical purity, and batch-specific characteristics before incorporating the material into experimental systems.
A typical Certificate of Analysis for research compounds may include the following analytical information:
- Identity confirmation using mass spectrometry or comparable analytical techniques appropriate for peptide verification
- Purity analysis performed using high-performance liquid chromatography (HPLC) or related chromatographic methods
- Molecular weight verification consistent with the expected peptide sequence
- Physical description of the compound, including the appearance of the lyophilized peptide powder
- Batch identification information such as lot number, manufacturing reference, and testing date
- Documentation of analytical methods used during internal quality verification procedures
Maintaining COA documentation allows researchers to track analytical characteristics associated with each compound lot used in laboratory experiments. These records support traceability of research materials and provide verification that analytical testing was performed prior to distribution. Proper documentation can also assist laboratories in maintaining accurate experimental records and supporting reproducibility across research projects.
Certificates of Analysis are available on the product pages or upon request, usually as per the most current lot tested.
Scientific References
- Advances in the detection of growth hormone releasing hormone synthetic analogs. Memdouh S, Gavrilović I, Ng K, Cowan D, Abbate V. Drug Testing and Analysis. 2021;13(11–12):1871–1887. https://doi.org/10.1002/dta.3183
- Growth hormone-releasing hormone receptor (GHRH-R) and its signaling. Halmos G, Szabo Z, Dobos N, Juhasz E, Schally AV. Reviews in Endocrine and Metabolic Disorders. 2025;26(3):343–352. Publication date: Feb 12, 2025 (Epub). https://doi.org/10.1007/s11154-025-09952-x
- DNA damage and growth hormone hypersecretion in pituitary somatotroph adenomas. Ben-Shlomo A, Deng N, Ding E, Yamamoto M, Mamelak A, Chesnokova V, Labadzhyan A, Melmed S. Journal of Clinical Investigation. 2020;130(11):5738–5755. Publication date: Nov 2, 2020. https://doi.org/10.1172/JCI138540
- Once-daily administration of CJC-1295, a long-acting growth hormone-releasing hormone (GHRH) analog, normalizes growth in the GHRH knockout mouse. Alba M, Fintini D, Sagazio A, Lawrence B, Castaigne JP, Frohman LA, Salvatori R. American Journal of Physiology – Endocrinology and Metabolism. 2006;291(6):E1290–E1294. Publication date: Jul 5, 2006 (Epub). https://doi.org/10.1152/ajpendo.00201.2006
- Activation of the GH/IGF-1 axis by CJC-1295, a long-acting GHRH analog, results in serum protein profile changes in normal adult subjects. Sackmann-Sala L, Ding J, Frohman LA, Kopchick JJ. Growth Hormone & IGF Research. 2009;19(6):471–477. Publication date: Apr 21, 2009 (Epub). https://doi.org/10.1016/j.ghir.2009.03.001
- Therapeutic Peptides in Orthopaedics: Applications, Challenges, and Future Directions. Rahman OF, Lee SJ, Seeds WA. Journal of the American Academy of Orthopaedic Surgeons Global Research & Reviews. 2026;10(1):e25.00236. Publication date: Jan 2, 2026. https://doi.org/10.5435/JAAOSGlobal-D-25-00236
- Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. Teichman SL, Neale A, Lawrence B, Gagnon C, Castaigne JP, Frohman LA. Journal of Clinical Endocrinology & Metabolism. 2006;91(3):799–805. Publication date: Dec 13, 2005 (Epub). https://doi.org/10.1210/jc.2005-1536
Regulatory Status & Legal Considerations (U.S.)
CJC-1295 No DAC is supplied strictly for laboratory research use only. This material is not approved by the U.S. Food and Drug Administration (FDA) for medical, diagnostic, or therapeutic use. It is intended solely for use by qualified professionals in controlled laboratory environments conducting in vitro studies, analytical testing, or preclinical research.
This product is not sold as a drug, dietary supplement, or food product, and it is not intended for human or veterinary use. Researchers and purchasing institutions are responsible for ensuring that all handling, storage, and experimental use of this compound complies with applicable federal, state, and institutional regulations governing laboratory research materials.
Additional information
| CAS | 863288-34-0 |
|---|---|
| PubChem CID | 91976842 |
| Molecular Weight | 3367.90 g/mol (CJC-1295 without DAC) |
| Amino Acid Sequence | Tyr-D-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg-NH2 |
| PubChem Link | |
| Product Note | For laboratory use only. Not for human or veterinary use. Proper handling and storage (-20°C) are required to maintain stability. Ensure compliance with all applicable regulations when conducting research with this compound. Peptides will arrive in a lyophilized (powder) form for maximum stability. |