GHK-Cu 100mg
$102.99
- 1 About GHK-Cu
- 2 GHK-Cu Mechanism of Action
- 3 GHK-Cu Research Value (Applications)
- 4 GHK-Cu vs BPC-157 vs TB-500 Comparison
- 5 GHK-Cu Storage & Handling Guide (Research Use Only)
- 6 Frequently Asked Questions (FAQ)
- 7 Certificate of Analysis (COA) & Quality Assurance
- 8 Scientific References
- 9 Regulatory Status & Legal Considerations (U.S.)
About GHK-Cu
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring copper-binding tripeptide complex formed when the endogenous peptide GHK coordinates with divalent copper ions (Cu²⁺). The peptide sequence Gly-His-Lys was originally identified in human plasma in the early 1970s during investigations into plasma-derived factors that influence cellular growth and extracellular matrix activity. Early work by Loren Pickart and colleagues helped characterize the peptide and its copper-binding properties, which quickly drew attention in biochemical and cell biology research.
Structurally, GHK is a short tripeptide capable of chelating copper through the histidine residue, forming the GHK-Cu complex that is frequently studied in experimental systems. This metal-peptide interaction enables the compound to participate in copper transport and metal-regulated signaling processes within biological models. Because copper ions play critical roles in enzymatic activity, oxidative balance, and connective tissue biology, GHK-Cu has become a useful model compound for studying peptide-mediated metal transport and copper-dependent cellular signaling pathways.
In laboratory settings, GHK-Cu is frequently investigated in cell culture assays, extracellular matrix signaling studies, inflammatory signaling models, and gene expression analyses related to peptide–metal interactions. Experimental systems have also explored how copper-binding peptides influence cellular responses involving oxidative stress regulation and tissue-associated signaling pathways in vitro and in animal models.
Compared to the endogenous tripeptide, synthetic GHK-Cu offers several advantages for laboratory research. Research Peptide’s rigorous, closely controlled synthesis provides consistent peptide identity and metal coordination, so that the complex demonstrates remarkable stability and reproducibility in assay systems to support reliable experimental design.
Research Peptides supplies research-grade GHK-Cu with analytical verification and batch-specific Certificates of Analysis, helping researchers maintain consistent experimental standards and reproducibility across studies.
GHK-Cu Mechanism of Action
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) functions primarily as a copper-binding signaling peptide complex that participates in cellular copper transport and copper-dependent regulatory pathways. In experimental systems, the complex acts as a carrier that facilitates copper ion availability to enzymes and signaling proteins involved in oxidative balance, extracellular matrix regulation, and gene expression.
These interactions have been investigated in cell culture systems, biochemical assays, and animal models, where GHK-Cu appears to influence multiple copper-sensitive molecular pathways. These findings are intended strictly for research contexts and should not be extended to any human or therapeutic application.
Primary Target Binding and Selectivity
Unlike receptor-specific peptide ligands, GHK-Cu operates primarily through metal coordination chemistry rather than classical receptor binding. The tripeptide sequence Gly-His-Lys forms a stable complex with divalent copper ions (Cu²⁺), largely through the imidazole group of histidine and additional coordination sites within the peptide backbone[1].
This chelation allows the complex to act as a biologically active copper carrier, capable of delivering copper to cellular proteins and enzymes that depend on trace metal cofactors for activity.
Studies in biochemical assays indicate that the GHK-Cu complex can interact with copper-dependent enzymes and transport systems, supporting copper availability within cellular environments[2]. Because copper ions participate in numerous enzymatic reactions, the peptide–metal complex has been examined as a model for studying regulated copper trafficking and peptide-mediated metal transport.
Downstream Signaling Pathways
Experimental studies suggest that copper delivery mediated by GHK-Cu may influence several intracellular signaling pathways associated with oxidative balance, transcriptional regulation, and extracellular matrix dynamics[3]. In cultured human fibroblasts and other cell models, exposure to GHK-Cu has been associated with changes in gene expression profiles related to cellular stress responses, growth signaling, and matrix-associated proteins[4].
Some laboratory investigations have reported involvement of pathways linked to MAPK/ERK signaling and transcriptional regulatory networks, which are commonly associated with cellular proliferation and differentiation processes in vitro[2]. These pathway interactions are typically evaluated using gene expression assays, protein phosphorylation measurements, and molecular signaling analyses in controlled experimental systems.
Functional Effects Observed in Preclinical Models
In preclinical research models, GHK-Cu has been studied for its effects on cellular growth markers, extracellular matrix protein expression, and oxidative stress indicators. In vitro experiments using fibroblast cultures have demonstrated shifts in transcriptional activity related to structural proteins and regulatory enzymes involved in tissue remodeling processes[2].
Animal studies have also explored how copper-binding peptides influence biomarker profiles associated with inflammatory signaling, antioxidant enzyme activity, and connective tissue metabolism. These observations are generally reported through biochemical endpoints such as gene expression changes, enzyme activity measurements, and histological analysis within controlled experimental models.
Pharmacology-Relevant Design Features
The small tripeptide structure of GHK contributes to its ability to rapidly coordinate copper ions and form a stable peptide–metal complex suitable for experimental applications. Compared to free copper ions, the peptide-bound form can provide controlled copper delivery within laboratory systems, allowing researchers to investigate copper-dependent processes with greater specificity.
Synthetic GHK-Cu used in laboratory studies offers consistent peptide identity and metal coordination, supporting reproducibility across experimental assays. Its relatively simple structure and stable copper-binding configuration make it a useful model compound for investigating metal-regulated signaling pathways and peptide-mediated trace element transport in preclinical systems.
GHK-Cu Research Value (Applications)
GHK-Cu is widely used in laboratory research as a model copper-binding peptide complex for studying metal-dependent cellular signaling and extracellular matrix regulation. Because copper ions are essential cofactors in numerous enzymatic and regulatory pathways, the GHK-Cu complex provides researchers with a controlled system for investigating peptide-mediated copper transport and copper-regulated biological processes.
GHK-Cu’s research applications span cell culture assays, biochemical pathway studies, and preclinical experimental models focused on oxidative balance, gene regulation, and connective tissue signaling.
Receptor/Pathway Mapping and Assay Development
Although GHK-Cu is not primarily a receptor-targeting peptide, it is frequently used in laboratory systems to examine metal-regulated signaling pathways and copper-dependent enzymatic processes. In vitro experiments often employ the peptide complex to study how controlled copper delivery influences intracellular signaling cascades and transcriptional activity in cultured cells[2].
Researchers have used GHK-Cu in cell-based assays to evaluate changes in gene expression, enzyme activation, and signaling protein phosphorylation associated with copper-dependent pathways[5]. Experimental systems may include fibroblast cultures, epithelial cell models, or other cell types where extracellular matrix dynamics and oxidative signaling can be measured.
Common analytical readouts include transcriptional profiling, reporter gene assays, enzyme activity measurements, and phosphorylation markers linked to pathways such as MAPK/ERK and other regulatory networks involved in cellular stress responses.
These controlled assay systems allow investigators to better understand how peptide-bound copper participates in molecular signaling and enzymatic regulation, helping refine experimental models of copper homeostasis and trace-metal–dependent biological processes.
Inflammation and Immune Signaling Research
Copper ions play recognized roles in immune signaling and oxidative stress regulation, making GHK-Cu a useful experimental tool for exploring peptide-mediated modulation of inflammatory pathways in laboratory models. Studies using cultured cells and animal systems have investigated how copper-binding peptides influence markers associated with immune signaling and cellular stress responses[6].
In vitro models often measure changes in cytokine expression profiles, oxidative stress indicators, and regulatory signaling proteins following exposure to copper-peptide complexes. These experiments may involve macrophage cultures, fibroblast models, or other cell systems used to evaluate inflammatory signaling pathways and immune-related gene expression.
Preclinical research has also explored how GHK-Cu influences biochemical markers associated with tissue remodeling and inflammatory regulation in animal models[2]. Observed endpoints may include shifts in enzyme activity, transcriptional responses, and other molecular indicators related to copper-dependent biological processes.
Such studies help researchers investigate how trace-metal transport and peptide signaling intersect with immune regulation and oxidative balance in controlled experimental environments.
Extracellular Matrix and Cellular Signaling Models
GHK-Cu is frequently studied in research focused on extracellular matrix regulation and connective tissue–associated cellular pathways[3]. Because copper-dependent enzymes participate in structural protein processing and matrix remodeling processes, the peptide–metal complex provides a useful experimental model for examining how trace elements influence cellular signaling in structural tissue environments.
In vitro investigations often evaluate gene expression patterns, structural protein synthesis markers, and enzymatic activity related to extracellular matrix dynamics. Fibroblast cultures are commonly used to measure transcriptional responses involving matrix-associated proteins, growth signaling regulators, and stress-response pathways.
Animal studies have also examined how copper-binding peptides affect biochemical markers associated with connective tissue metabolism and oxidative balance in preclinical models. These experiments typically assess measurable laboratory endpoints such as histological tissue markers, enzyme activity assays, and transcriptional changes within controlled research conditions[2].
Together, these experimental approaches help researchers explore the broader role of copper-regulated signaling in tissue-associated cellular processes.
| Note: GHK-Cu is supplied strictly for laboratory research use only. It’s not intended for human or veterinary use. |
GHK-Cu vs BPC-157 vs TB-500 Comparison
Peptides involved in cellular signaling, extracellular matrix regulation, and tissue-associated pathways are often studied together in experimental systems to understand differences in mechanism, stability, and model suitability. GHK-Cu is frequently examined alongside peptides such as BPC-157 and TB-500 (Thymosin Beta-4 fragment) because these compounds are commonly investigated in laboratory models focused on cell migration, oxidative signaling, and connective tissue–related pathways.
Comparing these peptides helps researchers evaluate differences in mechanistic targets, signaling behavior, and experimental design considerations across similar preclinical research domains.
| Field | GHK-Cu | BPC-157 | TB-500 (Thymosin Beta-4 Fragment) |
| Peptide Type | Copper-binding tripeptide complex | Synthetic gastric pentadecapeptide fragment | Synthetic fragment of thymosin beta-4 |
| Origin / Biological Context | Derived from endogenous human plasma peptide GHK that binds copper ions | Derived from a protective peptide sequence identified in gastric tissue | Fragment derived from the actin-regulating protein thymosin beta-4 |
| Primary Molecular Focus | Copper transport and copper-dependent enzymatic pathways | Nitric oxide signaling and vascular-associated pathways | Actin cytoskeleton regulation and cell migration pathways |
| Mechanism Summary | Chelates Cu²⁺ ions and facilitates controlled copper delivery that can influence gene expression and oxidative signaling in cellular systems | Investigated in models examining nitric-oxide–mediated signaling and vascular pathway activity | Interacts with actin-binding systems that regulate cytoskeletal organization and cellular motility |
| Typical Research Systems | Fibroblast cultures, extracellular matrix assays, oxidative stress models, rodent studies | Rodent tissue-injury models, vascular signaling assays, cell culture experiments | Cell migration assays, angiogenesis models, rodent tissue studies |
| Common Experimental Readouts | Gene expression profiling, oxidative stress markers, extracellular matrix protein expression | Nitric oxide markers, vascular signaling biomarkers, inflammatory pathway indicators | Cytoskeletal protein markers, migration assays, angiogenesis-related biomarkers |
| Stability in Experimental Systems | Metal-peptide complex provides controlled copper delivery in assay environments | Peptide fragment reported to demonstrate stability in several experimental models | Longer peptide fragment with activity associated with actin-regulating pathways in tissue models |
| Typical Research Domains | Extracellular matrix biology, oxidative signaling, metal-dependent pathways | Tissue signaling pathways, vascular biology, inflammatory signaling | Cell migration, cytoskeletal dynamics, tissue remodeling models |
| Key Study Design Tradeoffs | Useful for studying copper-mediated signaling but dependent on metal-peptide coordination conditions | Widely used in injury signaling models but mechanisms remain under active investigation | Frequently used in migration studies but broader signaling pathways may complicate interpretation |
| Disclaimer | For laboratory research use only. Not for human or veterinary use. | Same | Same |
GHK-Cu Storage & Handling Guide (Research Use Only)
GHK-Cu is supplied as a laboratory research compound and does not have a fully established clinical safety profile. The following guidance is intended strictly for controlled laboratory environments, where qualified personnel are operating under approved research protocols and institutional safety procedures. As such, all handling and experimental use should comply with institutional biosafety standards and applicable regulatory requirements.
Recommended laboratory best practices include:
- Follow institutional standard operating procedures (SOPs) and approved research protocols when handling peptide compounds.
- Wear appropriate personal protective equipment (PPE), including a laboratory coat, gloves, and eye protection, with additional controls used where required by institutional safety policies.
- Use appropriate engineering controls, such as a fume hood or biosafety cabinet, to minimize aerosolization or accidental exposure when preparing solutions or transferring materials.
- Store GHK-Cu under recommended cold conditions (typically −4 °F (−20 °C) for long-term storage) in a sealed, desiccated container protected from light to preserve compound stability.
- Avoid repeated freeze–thaw cycles, which may contribute to peptide degradation and reduced experimental consistency.
- Implement proper spill containment and laboratory waste disposal procedures consistent with institutional and federal chemical safety regulations.
- Maintain accurate documentation and traceability, including lot numbers and associated Certificates of Analysis (COAs), to support reproducibility and quality assurance in research workflows.
Like many peptide materials, GHK-Cu may demonstrate reduced stability in aqueous solution over time, particularly under conditions of elevated temperature or prolonged light exposure. Researchers commonly prepare solutions shortly before experimental use to maintain consistent assay conditions.
GHK-Cu is supplied strictly for laboratory research use only. It is not intended for human or veterinary use. Research Peptides does not provide medical, diagnostic, or therapeutic guidance.
Frequently Asked Questions (FAQ)
What is GHK-Cu used for in research?
GHK-Cu is commonly studied as a copper-binding peptide complex in laboratory models investigating cellular signaling, oxidative balance, and extracellular matrix–associated pathways. Researchers use the compound in cell culture systems, biochemical assays, and preclinical models to explore how peptide-mediated copper transport influences gene expression, enzyme activity, and trace-metal–dependent signaling networks.
How does GHK-Cu work at the molecular level?
In experimental systems, GHK-Cu functions primarily as a peptide–metal complex capable of binding and transporting copper ions (Cu²⁺). Copper is an essential cofactor in numerous enzymatic and regulatory processes. By coordinating copper through its Gly-His-Lys sequence, GHK-Cu can influence copper availability within cellular environments. Laboratory studies have explored how this interaction affects gene expression pathways, oxidative signaling networks, and extracellular matrix–related processes in vitro and in animal models.
How does GHK-Cu differ from other research peptides?
GHK-Cu differs from many peptide research compounds because its primary function involves metal coordination rather than receptor-specific signaling. While many peptides act as receptor agonists or antagonists, GHK-Cu operates through copper-binding chemistry, which enables it to participate in trace-metal transport and copper-dependent enzymatic systems. This makes the compound particularly useful in research examining metal-regulated cellular pathways, oxidative signaling, and extracellular matrix biology in controlled laboratory systems.
How should GHK-Cu be stored for research use?
GHK-Cu is typically supplied as a lyophilized peptide–metal complex to support stability during storage and transport. In laboratory settings, peptide materials are commonly stored under cold conditions such as −4 °F (−20 °C) for long-term storage, often in sealed, desiccated containers protected from light. Researchers should avoid repeated freeze–thaw cycles when preparing experimental solutions to maintain peptide integrity and reproducibility across assays.
How is the purity and identity of GHK-Cu verified?
Research-grade peptide materials are typically characterized using analytical techniques designed to confirm identity and composition before release for laboratory use. These methods often include high-performance liquid chromatography (HPLC) to assess purity and mass spectrometry to confirm molecular identity. Additional characterization may include molecular weight verification and appearance assessment of the lyophilized material.
What is the regulatory status of GHK-Cu?
GHK-Cu supplied by Research Peptides is designated strictly as a laboratory research compound. It is not approved as a drug, dietary supplement, or therapeutic product. The material is intended solely for in vitro experimentation, analytical research, or preclinical investigation conducted by qualified professionals in appropriate laboratory environments. Researchers are responsible for ensuring that all experimental work complies with institutional protocols and applicable regulatory standards.
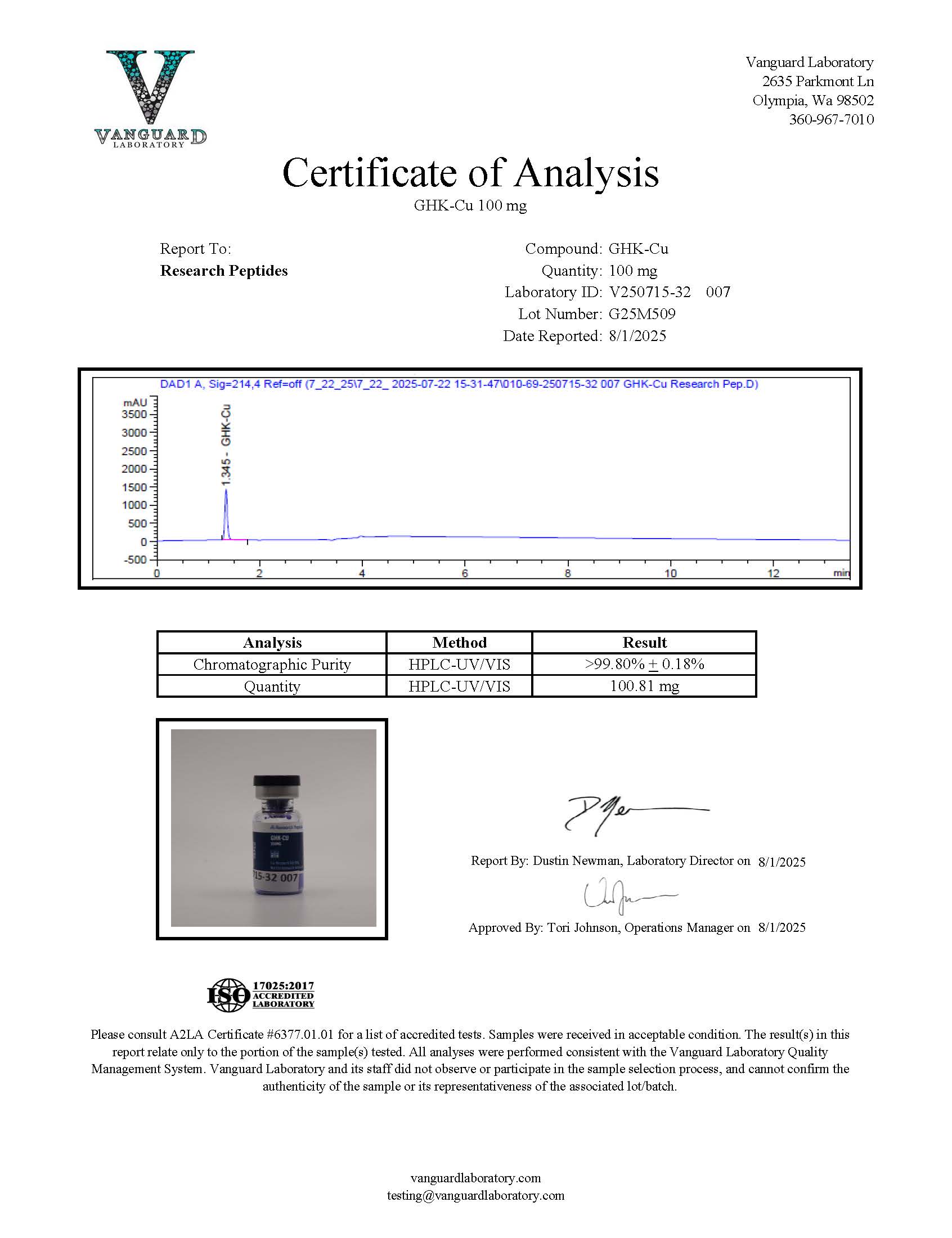
Certificate of Analysis (COA) & Quality Assurance
Each lot of GHK-Cu supplied by Research Peptides is accompanied by a Certificate of Analysis (COA) designed to support traceability, reproducibility, and transparency in laboratory research. The COA provides analytical documentation verifying key characteristics of the compound before it is released for distribution. These records help researchers confirm material identity and consistency when incorporating the compound into experimental workflows.
A typical COA for peptide research materials may include:
- Identity confirmation, such as mass spectrometry analysis or sequence verification methods appropriate for peptide characterization
- Purity assessment, commonly determined using high-performance liquid chromatography (HPLC) or comparable chromatographic techniques
- Relevant physicochemical characteristics, including molecular weight confirmation and appearance of the lyophilized material
- Lot number, testing date, and analytical method references used during quality evaluation
- Laboratory verification details, including documentation of independent or third-party analytical testing where applicable
Certificates of Analysis are available for review in PDF format on the product page or upon request, allowing researchers to verify analytical documentation associated with each product lot.
Scientific References
- The Human Tripeptide GHK-Cu in Prevention of Oxidative Stress and Degenerative Conditions of Aging: Implications for Cognitive Health. Pickart L, Vasquez-Soltero JM, Margolina A. *Oxidative Medicine and Cellular Longevity.* 2012;2012:324832. Published May 10, 2012.
https://doi.org/10.1155/2012/324832
- Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data, Pickart L, Margolina A. International Journal of Molecular Sciences, 19(7):1987, July 2018.
https://doi.org/10.3390/ijms19071987
- GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration, Pickart L, Vasquez-Soltero JM, Margolina A. *BioMed Research International,* 2015:648108, July 2015.
https://doi.org/10.1155/2015/648108
- The Effect of the Human Peptide GHK on Gene Expression Relevant to Nervous System Function and Cognitive Decline, Pickart L, Vasquez-Soltero JM, Margolina A. *Brain Sciences,* 7(2):20, February 2017.
https://doi.org/10.3390/brainsci7020020
- Exploring the Beneficial Effects of GHK-Cu on an Experimental Model of Colitis and the Underlying Mechanisms, Mao S, Huang J, Li J, Sun F, Zhang Q, Cheng Q, Zeng W, Lei D, Wang S, Yao J. Frontiers in Pharmacology, 16:1551843, July 2025.
https://doi.org/10.3389/fphar.2025.1551843
- Glycyl-l-histidyl-l-lysine prevents copper- and zinc-induced protein aggregation and central nervous system cell death in vitro. Jin-Hong Min, Heela Sarlus, Robert A Harris, , Metallomics, Volume 16, Issue 5, May 2024, mfae019. https://doi.org/10.1093/mtomcs/mfae019
Regulatory Status & Legal Considerations (U.S.)
GHK-Cu is supplied strictly for laboratory research use only. This material is not approved by the U.S. Food and Drug Administration (FDA) for medical, diagnostic, or therapeutic use. It is intended solely for use by qualified professionals in controlled laboratory environments conducting in vitro studies, analytical testing, or preclinical research.
This product is not sold as a drug, dietary supplement, or food product, and it is not intended for human or veterinary use. Researchers and purchasing institutions are responsible for ensuring that all handling, storage, and experimental use of this compound complies with applicable federal, state, and institutional regulations governing laboratory research materials.
Additional information
| CAS | 89030-95-5 |
|---|---|
| PubChem CID | 139063490 |
| Molecular Weight | 401.92 g/mol (GHK-Cu, copper-bound form) |
| Amino Acid Sequence | Gly-His-Lys |
| PubChem Link | |
| Product Note | For laboratory use only. Not for human or veterinary use. Proper handling and storage (-20°C) are required to maintain stability. Ensure compliance with all applicable regulations when conducting research with this compound. Peptides will arrive in a lyophilized (powder) form for maximum stability. |