Thymosin Beta-4 (TB-500) 10mg
$99.99
- 1 About TB-500 (Thymosin Beta-4)
- 2 TB-500 (Thymosin Beta-4) Mechanism of Action
- 3 TB-500 (Thymosin Beta-4) Research Applications
- 4 TB-500 vs Thymosin Beta-4 vs BPC-157 Comparison
- 5 TB-500 Storage & Handling Guide (Research Use Only)
- 6 Frequently Asked Questions (FAQ)
- 6.1 What purity standard does Research Peptides maintain for TB-500?
- 6.2 How is TB-500 typically reconstituted in laboratory research settings?
- 6.3 What storage conditions help maintain TB-500 peptide stability?
- 6.4 What makes TB-500 a useful research peptide for cytoskeletal signaling studies?
- 6.5 How does TB-500 differ from the full thymosin beta-4 peptide in research studies?
- 6.6 Why do researchers sometimes study TB-500 alongside peptides such as BPC-157?
- 6.7 What factors should researchers consider when selecting a TB-500 research peptide supplier?
- 7 Certificate of Analysis (COA) & Quality Assurance
- 8 Scientific References
- 9 Regulatory Status & Legal Considerations (U.S.)
About TB-500 (Thymosin Beta-4)
TB-500 is a synthetic research peptide derived from thymosin beta-4 (Tβ4), a naturally occurring actin-binding protein found in many mammalian tissues. In biological systems, thymosin beta-4 is involved in cellular structure regulation and signaling processes related to cytoskeletal dynamics. TB-500 represents a laboratory-produced analogue designed to replicate the key functional region of the larger protein, allowing researchers to study its biochemical behavior in controlled experimental environments.
Structurally, TB-500 corresponds to a peptide fragment associated with the active region of thymosin beta-4 responsible for interactions with actin and related cellular components and belongs to a class of small synthetic signaling fragments frequently used in molecular biology and cell culture experiments. By isolating this functional segment, researchers can investigate the molecular effects of thymosin-related signaling without relying on the full endogenous protein structure [1][2].
In laboratory research, TB-500 is commonly examined in models focused on actin polymerization, cellular migration pathways, angiogenic signaling processes, and gene expression patterns linked to cytoskeletal organization. These studies are typically conducted in cell culture systems or preclinical biochemical models designed to explore how peptides influence intracellular signaling networks, tissue remodeling mechanisms, and cellular response pathways.
Synthetic TB-500 offers several practical advantages over endogenously extracted protein in controlled research environments. Solid-phase synthesis enables precise, reproducible assembly of the peptide sequence, yielding a structurally consistent compound with defined molecular characteristics across batches. This consistency, combined with HPLC purification to ≥99% purity, minimizes batch-to-batch variability and reduces the risk of confounding results from structural irregularities or co-purified contaminants.
Synthetic production also confers improved stability under standard laboratory storage conditions, allowing the compound to achieve reliable performance across extended research timelines. These properties help researchers obtain reproducible experimental results while studying peptide-mediated signaling processes across different experimental conditions.
Purity and Quality Control
Each batch of TB-500 (10 mg) supplied by Research Peptides is verified through a dual analytical protocol prior to release. Analytical HPLC confirms the compound’s purity profile, which consistently achieves ≥99%, while mass spectrometry provides molecular weight confirmation and peptide sequence identity validation. Together, these methods ensure that every lot matches its expected molecular profile and meets the documentation standards required for rigorous laboratory research.
Batch-specific Certificates of Analysis are available for every order, providing transparent records of synthesis and analytical outcomes to support laboratory documentation and institutional verification requirements. As a dedicated U.S.-based supplier, Research Peptides maintains consistent manufacturing standards and reliable, secure domestic shipping to serve individual researchers, research laboratories, universities, and institutional procurement teams.
TB-500 (Thymosin Beta-4) Mechanism of Action
TB-500 functions primarily through interactions with cellular actin dynamics and cytoskeletal regulation pathways. As a synthetic analogue of the active region of thymosin beta-4, the peptide is investigated for its ability to influence actin polymerization, cell migration signaling, and intracellular structural organization. These mechanisms have been studied primarily in cell culture systems and preclinical experimental models examining cytoskeletal signaling and tissue remodeling processes.
Primary Target Binding and Selectivity
TB-500 is derived from the actin-binding region of thymosin beta-4, a protein known to interact with monomeric globular actin (G-actin). In experimental systems, the peptide is associated with modulation of actin sequestration and polymerization dynamics within the cytoskeleton. By interacting with actin monomers and associated regulatory proteins, TB-500 may influence the equilibrium between globular actin (G-actin) and filamentous actin (F-actin), a process central to cell structure, motility, and intracellular transport mechanisms [3].
These interactions are not mediated through a classical receptor binding site but rather through protein–protein interactions within cytoskeletal regulatory networks. As a result, TB-500 is frequently studied in molecular systems exploring how cytoskeletal remodeling affects cellular migration, structural integrity, and signaling coordination.
Downstream Signaling Pathways
Changes in cytoskeletal organization can influence multiple intracellular signaling pathways. Experimental research has shown that actin dynamics are closely linked to pathways such as MAPK/ERK and PI3K/AKT signaling, both of which participate in cellular growth regulation, gene transcription, and stress response signaling.
When cytoskeletal structure is altered through actin-binding activity, downstream signaling cascades may become modulated in cell-based assays. For example, changes in actin polymerization can influence focal adhesion complexes and integrin-mediated signaling, which in turn can activate kinase pathways that regulate transcription factors and cellular adaptation processes in laboratory models.
Functional Effects Observed in Preclinical Models
In preclinical studies and in vitro experiments, thymosin beta-4 and its active fragments have been associated with measurable effects on cellular migration, angiogenic signaling markers, and cytoskeletal organization. These observations are commonly reported in cell culture models examining endothelial cell movement, fibroblast activity, and molecular responses involved in tissue remodeling pathways [4].
Researchers frequently analyze TB-500 in assays evaluating changes in gene expression related to cytoskeletal proteins, extracellular matrix signaling components, and inflammatory signaling mediators. These experimental systems help investigators better understand how cytoskeletal regulators influence complex cellular behaviors within controlled research models.
Pharmacology-Relevant Design Features
Synthetic TB-500 provides researchers with a controlled and reproducible representation of the active signaling region of thymosin beta-4. Laboratory synthesis allows the peptide to maintain a consistent structure and purity profile, supporting reproducible experimental outcomes across different assay systems.
Compared with isolating the full endogenous protein, the smaller synthetic fragment offers advantages in stability, handling, and experimental reproducibility. These properties make TB-500 a useful molecular tool in studies investigating cytoskeletal signaling pathways, cellular migration processes, and peptide-mediated modulation of intracellular structural networks in preclinical research environments.
TB-500 (Thymosin Beta-4) Research Applications
TB-500 is a synthetic peptide research compound comprising the active region of thymosin beta-4 (Tβ4), an endogenous actin-binding peptide involved in cytoskeletal regulation, cell movement, and tissue organization. In published literature, a large share of the mechanistic work has been conducted on full-length thymosin beta-4 or its active actin-binding motif, while TB-500 is generally discussed as a synthetic fragment or analogue used to probe related biological processes in controlled experimental systems.
Therefore, TB-500 helps researchers separate direct compound-specific data from the broader thymosin beta-4 study landscape.
Researchers study TB-500 and related thymosin beta-4 fragments because they provide a practical way to investigate actin dynamics, cell migration behavior, angiogenic signaling, and inflammatory pathway modulation across multiple model systems. Depending on the research being conducted, the peptide may be evaluated in endothelial cell cultures, fibroblast assays, corneal epithelial systems, extracellular matrix experiments, transcriptional profiling workflows, or preclinical animal models.
In that sense, TB-500 peptide research is less about a single isolated pathway and more about how cytoskeletal control influences broader signaling networks.
In short, TB-500 study interest usually centers on a few recurring themes: how actin-binding motifs affect cell motility, which signaling pathways shift after cytoskeletal remodeling, how inflammatory transcription changes in stress models, and how peptide fragments behave in metabolism or stability experiments. Those are the domains where the compound has the most research relevance and where a well-characterized, analytically verified material is especially useful for reproducible work.
Actin Dynamics and Cytoskeletal Regulation
One of the most established areas of TB-500 research involves the regulation of actin cytoskeleton dynamics. Thymosin beta-4 is known to bind monomeric globular actin (G-actin), functioning as a major intracellular actin-sequestering peptide that regulates the equilibrium between globular actin and filamentous actin (F-actin). This equilibrium plays a fundamental role in cell structure, migration, and intracellular transport processes [4].
Experimental studies frequently examine TB-500 or thymosin beta-4 fragments in systems designed to analyze cytoskeletal organization. In fibroblast and epithelial cell models, researchers evaluate how actin-binding peptides influence actin polymerization rates, focal adhesion formation, and cell motility patterns. Techniques such as fluorescence microscopy, actin staining assays, and live-cell imaging are often used to visualize structural changes within the cytoskeleton.
Investigators may also measure downstream molecular markers associated with cytoskeletal remodeling. For example, cytoskeletal reorganization can influence integrin signaling complexes and focal adhesion kinase (FAK) activity, which subsequently affects intracellular pathways involved in cellular migration and adhesion. These experiments help clarify how actin-binding peptides regulate structural coordination within cells.
Angiogenesis and Endothelial Cell Signaling
Another prominent TB-500 study domain involves angiogenesis research. Thymosin beta-4 has been investigated extensively for its influence on endothelial cell migration, vascular sprouting, and signaling pathways associated with vascular remodeling in experimental systems [4].
Researchers commonly explore these mechanisms using endothelial cell cultures, aortic ring assays, or tube-formation models designed to simulate vascular network formation. Within these systems, investigators measure endpoints such as endothelial cell migration distance, tube formation capacity, nitric oxide signaling activity, and the expression of vascular growth markers.
Several signaling pathways appear to be associated with these processes in preclinical studies. Experimental work has linked thymosin beta-4 activity with pathways including PI3K/AKT and ERK/MAPK signaling cascades, both of which are known to regulate cell proliferation, survival signaling, and vascular remodeling responses in endothelial models [4]. By examining how actin-associated peptides influence these pathways, researchers gain insights into the molecular coordination between cytoskeletal organization and vascular signaling networks.
Cell Migration, Extracellular Matrix, and Remodeling Models
TB-500 peptide research is also frequently conducted in experimental systems focused on cellular migration and extracellular matrix interaction. Cell movement requires coordinated changes in cytoskeletal structure, adhesion complexes, and extracellular matrix signaling pathways, making actin-binding peptides relevant tools for studying these processes.
Laboratory models investigating this domain often involve scratch-wound assays, transwell migration experiments, or fibroblast matrix interaction studies. In these experiments, researchers evaluate how cytoskeletal signaling peptides influence the rate of cell migration across experimental surfaces or within matrix scaffolds.
Endpoints in these studies may include measurements of matrix metalloproteinase (MMP) expression, collagen deposition markers, and focal adhesion protein activity. These molecular indicators help researchers understand how cytoskeletal regulatory peptides affect extracellular matrix remodeling and tissue structural organization in controlled experimental environments.
Because extracellular matrix signaling interacts closely with cytoskeletal regulation, TB-500 signaling pathway research often intersects with integrin-mediated signaling networks and cell adhesion processes. These systems provide a useful framework for studying how peptide fragments derived from cytoskeletal proteins influence coordinated cell movement and structural adaptation.
Inflammatory Signaling and Transcriptional Response Research
Another area of active investigation involves inflammatory signaling pathways and transcriptional regulation. Thymosin beta-4 has been examined in various cell-based models exploring cytokine-driven inflammatory responses and transcription factor activation.
In epithelial and immune-adjacent cell culture systems, researchers analyze how thymosin-derived peptides influence signaling cascades such as NF-κB and MAPK pathways. These pathways regulate the expression of numerous cytokines and inflammatory mediators involved in cellular stress responses [2].
Experimental endpoints in these studies may include cytokine secretion profiles, gene transcription changes measured through RNA analysis, and phosphorylation patterns of signaling kinases. By monitoring these molecular indicators, investigators can explore how cytoskeletal peptides interact with broader cellular response networks under inflammatory stimulation conditions.
These transcriptional studies help clarify the relationship between cytoskeletal signaling and inflammatory pathway regulation, providing insight into how structural cellular proteins may influence gene expression programs during cellular stress responses.
Analytical and Metabolic Research Models
Beyond signaling research, TB-500 is also studied in analytical and metabolic experiments examining peptide stability, degradation pathways, and detection methods. Because TB-500 is a relatively short synthetic peptide fragment, researchers often investigate how it behaves in biological fluids or experimental assay systems.
Analytical chemistry techniques such as liquid chromatography–mass spectrometry (LC-MS) are commonly used to identify peptide metabolites and characterize degradation products in serum or enzyme-containing environments. These experiments help determine how peptide fragments break down, which metabolites appear during degradation, and how long the compound remains detectable in experimental systems.
This type of analytical work is particularly important for researchers conducting pharmacokinetic modeling in preclinical studies or validating experimental exposure levels in peptide assays. Understanding the metabolic profile of TB-500 helps ensure accurate interpretation of experimental outcomes in laboratory settings.
Because reproducibility is critical in peptide research, investigators often rely on analytically verified compounds with clearly documented purity and identity. Research Peptides provides TB-500 produced through advanced synthesis methods and verified through analytical testing, allowing researchers to work with a consistent compound profile when conducting signaling pathway experiments or biochemical studies.
| Note: TB-500 supplied by Research Peptides is intended strictly for laboratory research use only and is not approved for human or veterinary use. |
TB-500 vs Thymosin Beta-4 vs BPC-157 Comparison
Researchers studying cytoskeletal signaling peptides often evaluate TB-500 alongside closely related compounds that influence cellular migration, tissue remodeling pathways, and inflammatory signaling systems. TB-500 is derived from the active actin-binding motif of thymosin beta-4, while BPC-157 is a separate peptide frequently investigated in extracellular matrix and nitric-oxide–related signaling studies.
The comparison below highlights how these compounds differ in structure, signaling focus, and experimental applications in peptide research environments.
| Field | TB-500 | Thymosin Beta-4 (Tβ4) | BPC-157 |
| Compound Class | Synthetic peptide fragment derived from thymosin beta-4 | Naturally occurring endogenous peptide | Synthetic pentadecapeptide derived from gastric protein fragment |
| Structural Characteristics | Short acetylated heptapeptide fragment (Ac-LKKTETQ) | Full 43-amino-acid peptide protein | 15-amino-acid peptide fragment |
| Primary Biological Systems Studied | Cytoskeletal signaling and actin dynamics | Cytoskeletal organization, vascular signaling, and cell migration | Nitric oxide signaling, extracellular matrix biology, inflammatory pathways |
| Key Molecular Pathways Investigated | Actin polymerization equilibrium, focal adhesion signaling | PI3K/AKT, MAPK/ERK, endothelial signaling networks | NO signaling, VEGF-related pathways, inflammatory transcription signaling |
| Typical Experimental Models | Fibroblast migration assays, endothelial cell models, cytoskeletal imaging systems | Endothelial angiogenesis models, tissue remodeling experiments, developmental models | Fibroblast assays, inflammatory cell culture models, connective-tissue systems |
| Mechanistic Research Focus | Actin-binding motif behavior and cytoskeletal regulation | Broad signaling roles of thymosin beta-4 protein | Nitric-oxide pathway modulation and extracellular matrix signaling |
| Experimental Research Context | Used to isolate the active actin-binding region of thymosin beta-4 in signaling studies | Studied as the full endogenous regulatory peptide | Studied as a gastric-derived signaling peptide with distinct pathway interactions |
In experimental literature, TB-500 research is most closely associated with investigations of the actin-binding region of thymosin beta-4 and how cytoskeletal signaling influences cellular migration and structural organization. Because TB-500 represents a functional fragment of the larger protein, many TB-500 peptide study discussions appear alongside broader thymosin beta-4 signaling pathway research. Researchers sometimes examine both molecules within the same experimental framework to compare how the isolated actin-binding motif behaves relative to the full endogenous peptide.
By contrast, BPC-157 research generally explores different signaling domains. Studies on this peptide often focus on nitric-oxide-associated pathways, extracellular matrix signaling, and inflammatory transcription networks. While TB-500 peptide research and BPC-157 study literature may appear in similar experimental contexts—particularly cellular migration or connective-tissue models—their underlying molecular mechanisms differ substantially.
In some experimental designs, investigators studying structural peptides such as TB-500 also evaluate endocrine signaling peptides that influence systemic regulatory pathways. For example, growth hormone secretagogue research involving compounds such as Ipamorelin or GHRH analogs like CJC-1295 is frequently examined in parallel experimental discussions focused on cellular signaling, protein expression, and anabolic regulatory pathways.
While these peptides operate through entirely different biological systems, which are primarily the GH/IGF-1 endocrine axis rather than cytoskeletal signaling pathways, they are often explored alongside structural peptides in broader peptide research literature.
Understanding these distinctions helps researchers determine when TB-500 research is most appropriate for studying cytoskeletal dynamics and migration signaling, versus when endocrine-focused peptides such as Ipamorelin research or CJC-1295 peptide studies are better suited for experiments examining neuroendocrine signaling and growth hormone regulatory pathways.
TB-500 Storage & Handling Guide (Research Use Only)
TB-500 (Thymosin Beta-4) should be handled using the same precautions applied to other experimental peptide materials with limited clinical characterization. Proper storage, documentation, and preparation procedures help maintain compound integrity and reduce the risk of contamination or degradation. All handling should occur within controlled laboratory environments and follow institutional safety protocols and research procedures.
Standard Laboratory Handling Practices
Appropriate handling procedures help preserve peptide stability, maintain sample purity, and support safe laboratory operation when working with TB-500 in research environments.
- Follow institutional standard operating procedures (SOPs) and laboratory safety protocols when handling peptide compounds.
- Wear appropriate personal protective equipment (PPE), including laboratory coats, gloves, and protective eyewear.
- Prepare peptide solutions using sterile tools, calibrated pipettes, and clean laboratory equipment.
- Conduct preparation steps within appropriate engineering controls such as biosafety cabinets or chemical fume hoods when required by laboratory guidelines.
- Label all aliquots and working solutions clearly with compound name, concentration, preparation date, and lot number.
- Maintain accurate laboratory records documenting preparation methods, storage conditions, and experimental usage.
Consistent adherence to these procedures helps support reproducible research conditions and minimizes contamination risks during peptide preparation and handling.
Storage Guidelines for Compound Stability
Environmental factors such as temperature, moisture exposure, and repeated freeze–thaw cycles can influence peptide stability and experimental reliability. Controlled storage conditions help preserve the structural integrity of TB-500 for laboratory use.
- Store lyophilized TB-500 at −4°F (−20°C) for long-term storage.
- Store reconstituted peptide solutions at 36–46°F (2–8°C) when short-term storage is required for experimental use.
- Keep peptide vials tightly sealed and protected from moisture; desiccated storage is recommended for lyophilized materials.
- Avoid prolonged exposure to room temperature during preparation and handling procedures.
- Minimize repeated freeze–thaw cycles by preparing smaller aliquots of reconstituted peptide solutions.
- TB-500 is typically supplied as a lyophilized powder to improve stability prior to experimental preparation.
- Once reconstituted, peptide solutions may gradually degrade in aqueous environments due to hydrolysis or enzymatic activity, particularly at elevated temperatures.
Maintaining controlled storage and preparation conditions helps preserve peptide structure and supports consistent experimental performance in cytoskeletal and signaling pathway research models.
This compound is supplied strictly for laboratory research use only and is not intended for human or veterinary use. Research Peptides does not provide medical, diagnostic, or therapeutic guidance.
Frequently Asked Questions (FAQ)
What purity standard does Research Peptides maintain for TB-500?
TB-500 supplied by Research Peptides is manufactured to a purity level of ≥99%. Each batch undergoes analytical verification using high-performance liquid chromatography (HPLC) and mass spectrometry testing to confirm peptide identity and purity profile. These analytical methods help ensure sequence integrity and consistency, allowing researchers to work with a well-characterized TB-500 research peptide when conducting cytoskeletal or signaling pathway experiments.
How is TB-500 typically reconstituted in laboratory research settings?
TB-500 is commonly supplied as a lyophilized peptide powder for improved stability during storage and shipping. In laboratory environments, researchers typically prepare solutions by reconstituting the peptide using sterile water or bacteriostatic water under controlled conditions. The solvent is usually introduced slowly along the vial wall to minimize agitation, followed by gentle swirling to dissolve the peptide before aliquoting for experimental use.
What storage conditions help maintain TB-500 peptide stability?
Lyophilized TB-500 is generally stored at −4°F (−20°C) for long-term stability in laboratory storage environments. Once reconstituted, peptide solutions are typically refrigerated at 36–46°F (2–8°C) and used within a defined experimental timeframe. Researchers often prepare small aliquots to minimize repeated freeze–thaw cycles, which can contribute to peptide degradation in aqueous solution.
What makes TB-500 a useful research peptide for cytoskeletal signaling studies?
Researchers investigating TB-500 peptide research often focus on its relationship to the actin-binding region of thymosin beta-4. The peptide corresponds to a functional motif involved in cytoskeletal regulation, which makes it useful for studying actin dynamics, cell migration pathways, and focal adhesion signaling in experimental systems. As a result, TB-500 study literature frequently appears in research examining cellular migration assays, endothelial signaling models, and cytoskeletal pathway analysis.
How does TB-500 differ from the full thymosin beta-4 peptide in research studies?
TB-500 represents a short synthetic fragment derived from the actin-binding region of thymosin beta-4, while thymosin beta-4 itself is a 43–amino acid endogenous peptide found in many mammalian tissues. In research settings, TB-500 allows investigators to study the functional actin-interaction motif in isolation, whereas thymosin beta-4 research typically explores the broader signaling roles of the complete peptide protein.
Why do researchers sometimes study TB-500 alongside peptides such as BPC-157?
TB-500 and BPC-157 frequently appear together in peptide research discussions because both compounds are investigated in experimental models related to cellular migration and tissue remodeling pathways. However, the underlying signaling mechanisms differ. TB-500 research focuses primarily on cytoskeletal regulation and actin dynamics, whereas BPC-157 research often examines nitric oxide signaling, extracellular matrix pathways, and inflammatory transcription networks in experimental systems.
What factors should researchers consider when selecting a TB-500 research peptide supplier?
When evaluating a TB-500 research peptide, researchers typically consider analytical verification, peptide purity, and batch traceability. Suppliers that provide ≥99% purity verification, HPLC and mass spectrometry testing, and documented manufacturing standards help ensure that the compound used in experiments matches the expected molecular profile. These quality indicators support reproducibility when conducting TB-500 peptide research across different experimental systems.
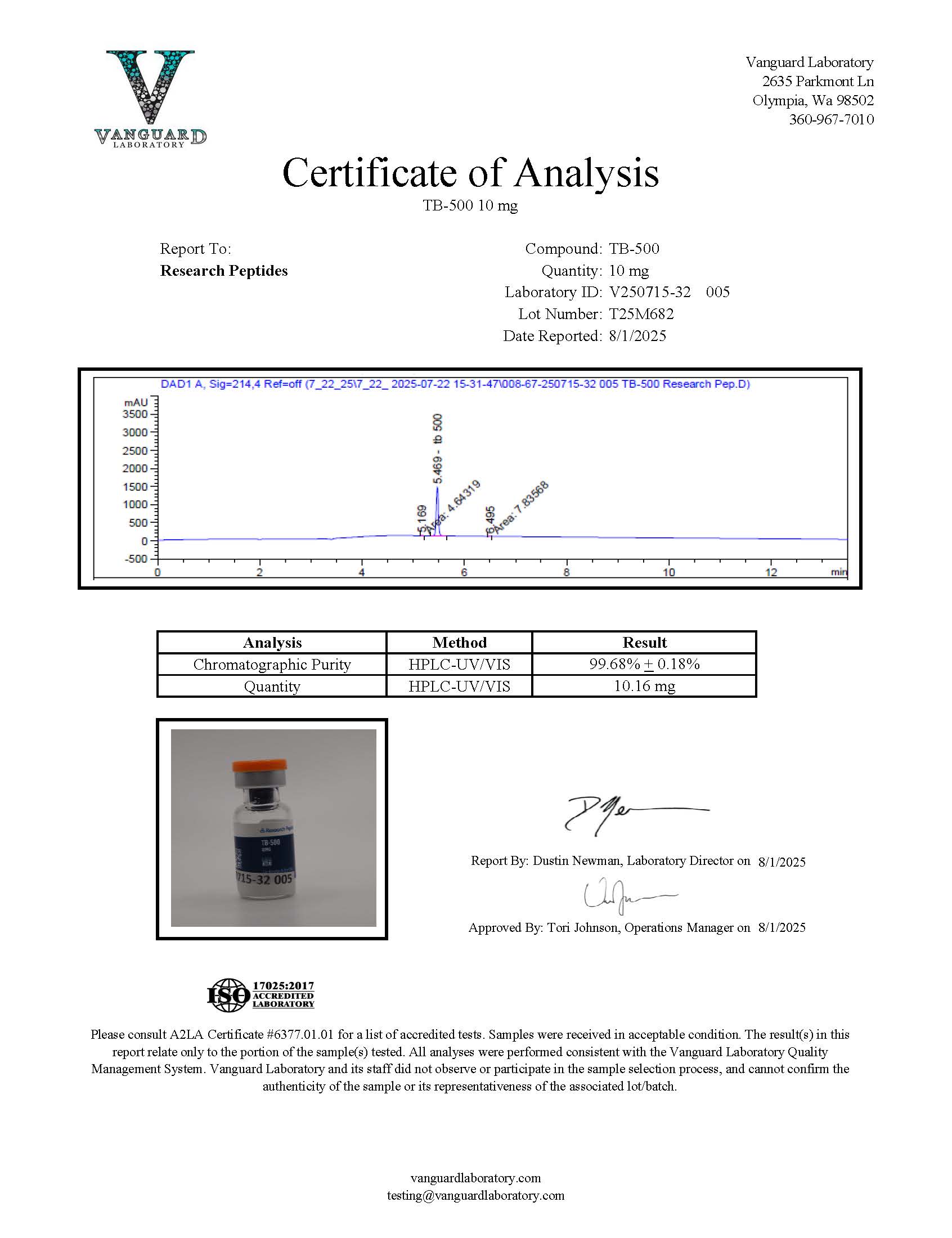
Certificate of Analysis (COA) & Quality Assurance
Each lot of TB-500 (Thymosin Beta-4) sold by Research Peptides is accompanied by a Certificate of Analysis (COA). This document provides detailed analytical information associated with the specific production batch and supports transparency in research compound sourcing. For peptide materials such as TB-500, analytical verification is conducted prior to distribution to confirm identity, purity characteristics, and batch-level consistency.
Maintaining batch-level analytical records allows researchers to reference documented testing results when preparing experimental materials. These records are commonly used to support laboratory documentation, experimental reproducibility, and internal quality review procedures within research environments.
The Certificate of Analysis serves as a formal record of the analytical testing performed during compound quality evaluation. Researchers may consult the COA to verify key analytical characteristics associated with the material, including compound identity, measured purity profile, and batch-specific testing data.
A typical Certificate of Analysis for research compounds may include the following analytical information:
- Identity confirmation using analytical techniques such as mass spectrometry to verify molecular weight and peptide sequence characteristics.
- Purity analysis performed using high-performance liquid chromatography (HPLC) or comparable chromatographic methods.
- Molecular weight verification consistent with the expected peptide structure.
- Physical description of the compound material, including the appearance of the lyophilized peptide powder.
- Batch identification information such as lot number, manufacturing reference, and analytical testing date.
- Documentation of internal analytical verification procedures conducted during quality evaluation.
COA documentation helps researchers maintain accurate laboratory records and supports traceability of experimental materials used in research protocols. By referencing batch-specific analytical data, investigators can verify the analytical testing associated with a compound lot and maintain documentation relevant to experimental reproducibility.
Certificates of Analysis are available on the product page when available, or request the documentation for the latest batch to review to help you buy TB-500 with confidence.
Scientific References
- Thymosin β4 and thymosin β4-derived peptides induce mast cell exocytosis. Janina Wyczółkowska, Aurelia Walczak-Drzewiecka, Waldemar Wagner, Jarosław Dastych. Peptides. 2007;28(4):752–759. https://doi.org/10.1016/j.peptides.2007.01.004
- Utilizing Developmentally Essential Secreted Peptides Such as Thymosin Beta-4 to Remind the Adult Organs of Their Embryonic State—New Directions in Anti-Aging Regenerative Therapies. Maar K, Hetenyi R, Maar S, Faskerti G, Hanna D, Lippai B, Takatsy A, Bock-Marquette I. Cells. 2021;10(6):1343. Publication date: May 28, 2021. https://doi.org/10.3390/cells10061343
- Structural basis of thymosin-β4/profilin exchange leading to actin filament polymerization. Xue B, Leyrat C, Grimes JM, Robinson RC. Proceedings of the National Academy of Sciences of the United States of America. 2014;111(43):E4596–E4605. Publication date: October 28, 2014. https://doi.org/10.1073/pnas.1412271111
- Progress on the Function and Application of Thymosin β4. Xing Y, Ye Y, Zuo H, Li Y. Frontiers in Endocrinology. 2021;12:767785. Publication date: December 21, 2021. https://doi.org/10.3389/fendo.2021.767785
- Thymosin beta 4: A novel corneal wound healing and anti-inflammatory agent. Sosne G, Qiu P, Kurpakus-Wheater M. Clinical Ophthalmology. 2007;1(3):201–207. Not listed.
Regulatory Status & Legal Considerations (U.S.)
TB-500 peptide is supplied strictly for laboratory research use only. This material is not approved by the U.S. Food and Drug Administration (FDA) for medical, diagnostic, or therapeutic use. It is intended solely for use by qualified professionals in controlled laboratory environments conducting in vitro studies, analytical testing, or preclinical research.
This product is not sold as a drug, dietary supplement, or food product, and it is not intended for human or veterinary use. Researchers and purchasing institutions are responsible for ensuring that all handling, storage, and experimental use of this compound complies with applicable federal, state, and institutional regulations governing laboratory research materials.
Additional information
| CAS | 77591-33-4 |
|---|---|
| PubChem CID | 16132341 |
| Molecular Weight | 4963.49 g/mol |
| Amino Acid Sequence | Ac-Ser-Asp-Lys-Pro-Asp-Met-Ala-Glu-Ile-Glu-Lys-Phe-Asp-Lys-Ser-Lys-Leu-Lys-Lys-Thr-Glu-Thr-Gln-Glu-Lys-Asn-Pro-Leu-Pro-Ser-Lys-Glu-Thr-Ile-Glu-Gln-Glu-Lys-Gln-Ala-Gly-Glu-Ser |
| PubChem Link | |
| Product Note | For laboratory use only. Not for human or veterinary use. Proper handling and storage (-20°C) are required to maintain stability. Ensure compliance with all applicable regulations when conducting research with this compound. Peptides will arrive in a lyophilized (powder) form for maximum stability. |